
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
SCHEDULE 14A
Proxy Statement Pursuant to Section 14(a) of the
Securities Exchange Act of 1934
Filed by the Registrant x☒
Filed by a Party other than the Registrant ¨☐
Check the appropriate box:
☐ | ||
Preliminary Proxy Statement | ||
☐ | Confidential, for Use of the Commission Only (as permitted by Rule 14a-6(e)(2)) | |
☒ | Definitive Proxy Statement | |
☐ | Definitive Additional Materials | |
☐ | Soliciting Material under Rule 14a-12 |
PUMA BIOTECHNOLOGY, INC.
(Name of the Registrant as Specified In Its Charter)
(Name of Person(s) Filing Proxy Statement, if other than the Registrant)
Payment of Filing Fee (Check the appropriate box)all boxes that apply):
☒ | |||||
No fee required. | |||||
| |||||
| |||||
| |||||
| |||||
| |||||
| Fee paid previously with preliminary materials. | |||||
☐ | Fee computed on table in exhibit required by Item 25(b) per Exchange Act Rules 14a-6(i)(1) and 0-11. | ||||
| |||||
| |||||
| |||||
| |||||

April 26, 2024

April 29, 2016
Fellow Stockholder:
You are invited to attend the annual meeting of stockholders of Puma Biotechnology, Inc. (the “Company,” “we,” “us” or “our”) to be held on Monday,Tuesday, June 13, 2016,18, 2024, at 1:00 p.m. local time, at the Luxe Sunset Boulevard Hotel, 11461 Sunset Boulevard,Company’s principal executive offices, 10880 Wilshire Blvd., Suite 2150, Los Angeles, CA 90049.90024.
At this year’s annual meeting you will be asked to:
1. | Elect eight directors to serve for a one-year |
2. | Ratify the selection of KPMG LLP as our independent registered public accounting |
3. | Vote on an advisory basis to approve the compensation of our named executive officers as described in the proxy statement (“say-on-pay vote”); |
| 4. | Vote on an advisory basis regarding the frequency of future say-on-pay votes (“frequency vote”); |
| 5. | Approve an amendment to increase the number of shares of the Company’s common stock reserved for issuance under the Puma Biotechnology, Inc. |
6. |
| Transact such other business as may properly come before the annual meeting. |
The accompanying Notice of Annual Meeting and proxy statement describe these matters. We urge you to read this information carefully.
The Board of Directors unanimously believes that election of its nominees to serve as our directors, ratification of our independent registered public accounting firm, and approval of amendments to the Certificate of Incorporation to eliminate the ability of stockholders to act by written consent and to eliminate the mandatory indemnification of all persons covered by Section 145 of the General Corporation Law of the State of Delawaresay-on-pay vote, are in the best interests of the Company and its stockholders, and accordingly, recommends a vote “FOR” each of the eight nominees for director named in the proxy statement, a vote “FOR” the ratification of KPMG LLP as our independent registered public accounting firm, a vote “FOR” the say-on-pay vote, a vote for “1 Year” with respect to the frequency vote and a vote “FOR” each of the other proposals identified above.2011 Plan Amendment.
It is important that your shares be represented and voted whether or not you plan to attend the annual meeting in person. You may submit your proxy over the Internet, or if you are receiving a paper copy of the proxy statement, by telephone or by completing and mailing athe proxy card.card sent with the proxy statement. Submitting your proxy over the Internet, by telephone or by written proxy will ensure your shares are represented at the annual meeting.
The Board of Directors appreciates and encourages stockholder participation. Thank you for your continued support.
|
 |
Alan H. Auerbach Chairman, President, Chief Executive Officer and Secretary |
| 43 | ||||
| 43 | ||||
| 43 | |||
Appendix A Second Amended and Restated Certificate of Incorporation


PUMA BIOTECHNOLOGY, INC.
10880 Wilshire Boulevard, Suite 2150,
Los Angeles, California 90024
NOTICE OF ANNUAL MEETING OF STOCKHOLDERS
TO BE HELD ON MONDAY,TUESDAY, JUNE 13, 201618, 2024
To the Stockholders of Puma Biotechnology, Inc. (the “Company,” “we” and “our”):
We will hold an annual meeting of stockholders of the Company at the Luxe Sunset Boulevard Hotel, 11461 Sunset Boulevard,our principal executive offices, located at 10880 Wilshire Blvd., Suite 2150, Los Angeles, California 90049,CA 90024, on Monday,Tuesday, June 13, 2016,18, 2024, at 1:00 p.m. local time. At the annual meeting we will consider and act upon the following matters:
1. | Election of |
2. | Ratification of the selection of |
3. | Advisory (non-binding) vote to approve the compensation of |
| 4. |
5. | Approve an amendment to increase the |
6. | Such other business as may properly come before the annual meeting or any adjournments or postponements of the annual meeting. |
The proxy statement accompanying this notice describes each of these items of business in detail. The Board of Directors recommends a vote “FOR” each of the eight nominees for director named in the proxy statement, a vote “FOR” the ratification of the selection of PKF Certified Public AccountantsKPMG LLP as our independent registered public accounting firm, a vote “FOR” the say-on-pay vote, a vote for “1 Year” for the frequency vote and a vote “FOR” the approval of each of the proposed amendments to the Certificate of Incorporation.2011 Plan Amendment.
Only the Company’s stockholders of record at the close of business on April 20, 2016,22, 2024, the record date for the determination of stockholders entitled to notice of and to vote at the annual meeting, or any adjournmentadjournments or postponementpostponements thereof, are entitled to notice of, and to vote at, the annual meeting. On April 20, 2016,22, 2024, we had 32,493,09248,238,850 shares of common stock outstanding. A list of stockholders eligible to vote at the annual meeting will be available for inspection at the annual meeting, and at the Company’s principal executive offices at 10880 Wilshire Boulevard, Suite 2150, Los Angeles, CA 90024 during regular business hours for a period of no less than ten days prior to the annual meeting. Please contact Investor Relations at (424) 248-6500 or ir@pumabiotechnology.com if you would like to visit our offices to review the stockholder list.
Your vote is very important.It is important that your shares be represented and voted whether or not you plan to attend the annual meeting in person. If you are viewing the proxy statement on the Internet, you may grant your proxy electronically via the Internet by following the instructions on the Notice of Internet Availability of Proxy Materials previously mailed to you and the instructions listed on the Internet site. If you are receiving a paper copy of the proxy statement, you may submit your proxy by completing and mailing the proxy card enclosed with the proxy statement, or you may grant your proxy electronically via the Internet or by telephone by following the instructions on the proxy card.card provided to you. Submitting a proxy over the Internet, by telephone or by mailing a proxy card will ensure your shares are represented at the annual meeting.
The Luxe Sunset Boulevard Hotelannual meeting is accessible to those who require special assistance or accommodation. If you require special assistance or accommodation, please contact Investor Relations at (424) 248-6500 or ir@pumabiotechnology.com or write to: Puma Biotechnology, Inc., 10880 Wilshire Boulevard, Suite 2150, Los Angeles, California 90024, Attention: Investor Relations.
By Order of the Board of Directors, |
 |
|
| Alan H. Auerbach |
Chairman, President, Chief Executive Officer and Secretary |
INFORMATION CONCERNING VOTING AND SOLICITATION
Your proxy is being solicited on behalf of the Board of Directors (the “Board”) of Puma Biotechnology, Inc., a Delaware corporation (the “Company,” “we,” “us” or “our”), for use at our 20162024 annual meeting of stockholders to be held on Monday,Tuesday, June 13, 2016,18, 2024, at 1:00 p.m. local time, at the Luxe Sunset Boulevard Hotel, 11461 Sunset Boulevard,Company’s principal executive offices, 10880 Wilshire Blvd., Suite 2150, Los Angeles, California 90049,CA 90024, or at any continuation, postponement or adjournment thereof (the “annual meeting”), for the purposes discussed in this proxy statement and in the accompanying Notice of Annual Meeting and any other business properly brought before the annual meeting. Proxies are solicited to give all stockholders ofwho held shares on the record date an opportunity to vote on matters properly presented at the annual meeting.
In accordance with the Securities and Exchange Commission’s “notice and access” model, we have elected to provide access to our proxy materials, including our notice of annual meeting, this proxy statement and our annual report to stockholders, over the Internet. Accordingly, onOn or about April 29, 2016,26, 2024, we intend to make our proxy materials available on the Internet and to mail a Notice of Internet Availability of Proxy Materials (the “Notice”) to all of our stockholders as of record. On or about our record date on April 29, 2016, we also22, 2024. We intend to mail a paper copy of the proxy materials and proxy card to other stockholders of record who have elected to receive such materials in paper form.form on or about April 26, 2024. Brokers and other nominees who hold shares on behalf of beneficial stockholders will be sending their own similar notice.Notice to such beneficial owners. All stockholders will have the ability to access the proxy materials on the website referred to in the Notice or to request to receive a printed set of the proxy materials. Instructions on how to request a printed copy by mail or electronically may be found on the Notice and on the website referred to in the Notice, including an option to request paper copies on an ongoing basis. If you properly request a printed copy of the proxy materials, we intend to mail the proxy materials, together with a proxy card, to you, within three business days of such request.
Important Notice Regarding the Availability of Proxy Materials for the 20162024 Annual Meeting of Stockholders to be Held on Tuesday, June 13, 201618, 2024
The Notice of Annual Meeting, this proxy statement, proxy card sample and our 20152023 Annual Report, which consists of a letter to stockholders and our Annual Report on Form 10-K for the fiscal year ended December 31, 2015,2023, are available on our website athttp:https://investor.pumabiotechnology.com/annual-meetingsec-filings/annual-reports-and-proxies/default.aspx. This website address contains the following documents: the Notice, the proxy statement and proxy card sample, and the 2015 Annual Report. You are encouraged to access and review all of the important information contained in the proxy materials before voting.
You are entitled to vote at the annual meeting if you were a stockholder of recordholder of our common stock as of the close of businessbusiness on April 20, 2016. 22, 2024. You are entitled to one vote for each share of common stock held on all matters to be voted upon at the annual meeting. Your shares may be voted at the annual meeting only if you are present in person or represented by a valid proxy.
You may vote by attending the annual meeting and voting in person or you may submitsubmitting a proxy to have your shares voted at the annual meeting.meeting, or by attending the annual meeting and voting in person. The method of submitting your proxy will differ depending on whether you are viewing this proxy statement on the Internet or receiving a paper copy and whether you are a beneficial stockholder or a stockholder of record.
Beneficial Stockholders. Beneficial stockholders hold their shares through a broker, bank, trustee or other nominee (that(“broker”) that is in “street name”) rather than directly in their own name. If you hold your shares in street name,
you are a “beneficial stockholder,” and the Notice and proxy materials wereare made available to you by the organizationbroker holding your account. This organizationshares. Your broker is considered the stockholder of record for purposes of voting at the annual meeting. As a beneficial stockholder, you have the right to instruct that organizationyour broker on how to vote the shares held in your account. If you requested printed copies of the proxy materials by mail, you will receive a voting instruction form from your bank, broker, trustee or other nominee.
Stockholders of Record. If your shares are registered directly in your name with our transfer agent, Wells Fargo Bank, N.A.,EQ Shareowner Services, or if you hold stock certificates in your name, you are considered the stockholder of record with respect to those shares, and the Notice and proxy materials wereare made available directly to you by the Company.us. If you requestedare receiving printed copies of the proxy materials by mail, you will receive a proxy card from us.
Voting/Submitting Proxy. Whether you are a stockholder of record or a beneficial stockholder, you may direct how your shares are voted without attending the annual meeting. If you are a stockholder of record, you may submit a proxy to authorize how your shares are voted at the annual meeting. You can submit a proxy over the Internet by following the instructions on the website referred to in the Notice or, if you requested and received printed copies of the proxy materials, you can also submit a proxy by mail or telephone pursuant to the instructions on the proxy card enclosed with the proxy materials.
If you are a beneficial stockholder, you may also submit your voting instructions to the broker holding your shares over the Internet by following the instructions provided in the Notice, or, if you requested and received printed copies of the proxy materials, you can also submit voting instructions by telephone or mail by following the instructions provided to you by your bank, broker, trustee or other nominee.broker.
Submitting your proxy or voting instructions via the Internet, by telephone or by mail will not affect your right to vote in person should you decide to attend the annual meeting, although beneficial stockholders must obtain a “legal proxy” from the bank,your broker trustee or other nominee that holds their shares giving themyou the right to vote the shares at the annual meeting in order to vote in person at the meeting.
The Internet and telephone voting facilities will close at 12:00 noon (CT)11:59 p.m. Eastern Time on June 12, 2016.17, 2024. If you vote through the Internet, you should be aware that you may incur costs to access the Internet, such as usage charges from telephone companies or Internet service providers and that these costs must be borne by you. If you submit your proxy over the Internet or by telephone, then you do not need notto return a written proxy card by mail. If you intendYou will need the 16 digit control number contained on the Notice, or proxy card, as applicable, to submit your proxyvote over the Internet or by telephone and have not done so prior to 12:00 noon (CT) on June 12, 2016, your only alternative if you wish to vote at the annual meeting will be to attend the annual meeting and vote in person.telephone.
YOUR VOTE IS VERY IMPORTANT.You should submit your proxy even if you plan to attend the annual meeting. If you properly give your proxy and submit it to us in time to vote, one of the individuals named as your proxy will vote your shares as you have directed.
All shares entitled to vote and represented by properly submitted proxies (including those submitted electronically, telephonically and in writing) that have not been properly revoked, will be voted at the annual meeting in accordance with the instructions indicated on those proxies. If no direction is indicated on a properly submitted proxy, your shares will be voted “FOR” each of the eight nominees for director named in the proxy statement, “FOR” the ratification of the selection of PKF Certified Public AccountantsKPMG LLP (“KPMG”) as our independent registered public accounting firm, “FOR” the say-on-pay vote, for “1 Year” for the frequency vote and “FOR” the approval of the amendments to the Company’s Amended and Restated Certificate of Incorporation (the “Certificate of Incorporation”) to eliminate the ability of stockholders to act by written consent and to eliminate the mandatory indemnification of all persons covered by Section 145 of the General Corporation Law of the State of Delaware.2011 Plan Amendment. The proxy gives each of Alan H. Auerbach and Charles R. EylerMaximo F. Nougues discretionary authority to vote your shares in accordance with his best judgment with respect to all additional matters that might properly come before the annual meeting.
If you are a stockholder of record, you may revoke your proxy at any time before your proxy is voted at the annual meeting by taking any of the following actions:actions before the annual meeting:
delivering to our Corporate Secretary a signed written notice of revocation, bearing a date later than the date of the proxy, stating that the proxy is revoked;
• | delivering to our Corporate Secretary a signed written notice of revocation, bearing a date later than the date of the proxy, stating that the proxy is revoked; |
signing and delivering a new paper proxy, relating to the same shares and bearing a later date than the original proxy;
• | signing and delivering a new paper proxy, relating to the same shares and bearing a later date than the original proxy; |
submitting another proxy by telephone or over the Internet (the proxy holders will vote your shares in accordance with your latest telephone or Internet voting instructions); or
• | submitting another proxy by telephone or over the Internet (the proxy holders will vote your shares in accordance with your latest timely-submitted telephone or Internet voting instructions); or |
• | attending the annual meeting and voting in person, although attendance at the annual meeting will not, by itself, revoke a proxy. |
attending the annual meeting and voting in person, although attendance at the annual meeting will not, by itself, revoke a proxy.
Written notices of revocation and other communications with respect to the revocation of proxies should be addressed to:
Puma Biotechnology, Inc.
10880 Wilshire Boulevard, Suite 2150
Los Angeles, CA 90024
Attention: Corporate Secretary
If you are a beneficial stockholder, and you submit a voting instruction form, you may change your vote by submitting new voting instructions to your bank, broker trustee or other nominee in accordance with the procedures of such bank, broker, trustee or other nominee.broker.
If you plan to attend the annual meeting and wish to vote in person, you will be given a ballot at the annual meeting. Beneficial stockholders must obtain and present at the annual meeting a “legal proxy” from the broker that holds their shares giving them the right to vote the shares at the annual meeting in order to vote in person at the meeting.
At the close of business on April 20, 2016, 32,493,092 22, 2024, 48,238,850 shares ofof our common stock were outstanding and entitled to vote.vote at the annual meeting. All votes will be tabulated by the inspector of election appointed for the annual meeting, who will separately tabulate votes “for,” “against,” abstentions and broker non-votes.meeting.
Quorum.A majority of the outstanding shares of common stock, present in person or represented by proxy, will constitute a quorum at the annual meeting. Shares of common stock held by persons attending the annual meeting but not voting, shares represented by proxies that reflect abstentions as to a particular proposal and broker “non-votes,” if any,“non-votes” will be counted as present for purposes of determining a quorum.
Broker Non-Votes. Brokers or other nominees who hold shares of common stock in “street name” for a beneficial owner of those shares typically have the authority to vote in theirthe broker’s discretion on “routine” proposals when they have not received instructions from beneficial owners.owners on how to vote on such proposal. However, brokers are not allowed to exercise their voting discretion with respect to the election of directors or for the approval of certain “non-routine” matters,proposals without specific instructions from the beneficial owner. These non-voted shares are referred to as “broker non-votes.” Only Proposal 2 (ratifying the appointment of our independent registered public accounting firm), Proposal 3 (amendment of Certificate of Incorporation to eliminate the ability of stockholders to act by written consent) and Proposal 4 (amendment of Certificate of Incorporation to eliminate the mandatory indemnification of all persons covered by Section 145 of the General Corporation Law of the State of Delaware) are is considered a routine matters.matter. If you are a beneficial stockholder holding shares through a broker or other nominee and you do
not submit instructions on how your shares should be voted, your broker or other nominee will not be able to vote your shares on Proposal 1 (election of directors)., Proposal 3 (say-on-pay vote), Proposal 4 (frequency vote) and Proposal 5 (2011 Plan Amendment) and broker non-votes will result on these proposals.
Votes Required
Proposal 1 – Election of Directors. Directors will be elected by a plurality of the votes of the shares present in person or represented by proxy and entitled to vote.vote on the election of directors. Stockholders will be given the choice to vote “for” or “withhold” votes for each nominee. Thus, the fiveeight nominees receiving the greatest number of votes “FOR” their election will be elected. Abstentions and brokerBroker non-votes do not represent “for” or “withhold” votes as they are not considered votes castentitled to vote, and therefore broker non-votes will not affect the outcome of the vote.vote for this proposal.
Proposal 2 – Ratification of Independent Registered Public Accounting Firm.The affirmative vote of a majority of the votes cast at the annual meetingon this proposal is required for the ratification of the selection of PKF Certified Public Accountants, a Professional Corporation,KPMG as our independent registered public accounting firm for the year ending December 31, 2016.2024. Stockholders will be given the choice to vote “for” or “against” or “abstain” on this proposal. Abstentions are not considered votes cast and therefore will not affect the outcome of the vote. Brokers have discretionary authority in the absence of timely instructions from their beneficial owners to vote on this proposal. As a result, therebroker non-votes are not expected on this proposal.
Proposal 3 – Advisory Say-on-Pay Vote. The affirmative vote of a majority of the shares cast on this proposal is required for approval, on an advisory basis, of the compensation of our named executive officers as disclosed in the proxy statement. Stockholders will be nogiven the choice to vote “for” or “against” or “abstain” on this proposal. Abstentions and broker non-votes are not considered votes cast and therefore will not affect the outcome of the vote for this proposal.
Proposal 4 – Advisory Frequency Vote. The affirmative vote of a majority of the shares cast on this proposal is required for approval, on an advisory basis, of the frequency of future say-on-pay votes. Stockholders will be given the choice to vote “for” or “against” or “abstain” on this proposal. Abstentions and broker non-votes are not considered votes cast and therefore will not affect the outcome of the vote. With respect to this item, if none of the frequency alternatives (1 year, 2 years or 3 years) receives a majority of votes cast, we will consider the frequency that receives the highest number of votes by stockholders to be the frequency that has been selected by our stockholders. However, because this vote is advisory and not binding on us or our Board in any way, our Board may decide that it is in our and our stockholders’ best interests to hold a say-on-pay vote more or less frequently than the option approved by our stockholders.
Proposal 5– Approval of Amendment to Certificate of Incorporation to Eliminate the Ability of Stockholders to Act by Written Consent.2011 Plan Amendment. The affirmative vote of a majority of the shares issued and outstanding as ofvotes cast at the record dateannual meeting is required for the approval of this amendmentthe 2011 Plan Amendment. Stockholders will be given the choice to the Certificate of Incorporation. Abstentions will have the same effect as votes against this proposal. Brokers have authority in the absence of timely instructions from their beneficial owners to vote “for” or “against” or “abstain” on this proposal. As a result, there will be noAbstentions and broker non-votes are not considered votes cast and therefore will not affect the outcome of the vote for this proposal.
Proposal 4 – Approval of Amendment to Certificate of Incorporation to Eliminate the Mandatory Indemnification of All Persons Covered by Section 145 of the General Corporation Law of the State of Delaware. The affirmative vote of a majority of the shares issued and outstanding as of the record date is required for approval of this amendment to the Certificate of Incorporation. Abstentions will have the same effect as votes against this proposal. Brokers have authority in the absence of timely instructions from their beneficial owners to vote on this proposal. As a result, there will be no broker non-votes for this proposal.
Our Board is soliciting proxies for the annual meeting from our stockholders. We will bear the entire cost of soliciting proxies from our stockholders. In addition to the solicitation of proxies by delivery of the Notice or proxy statement by mail, we will request that brokers banks and other nominees that hold shares of our common stock, which are beneficially owned by our stockholders, send Notices, proxies and proxy materials to those beneficial owners and secure those beneficial owners’ voting instructions. We will reimburse those record holdersthe brokers for their reasonable expenses. We have engaged Innisfree M&A Incorporated to assist in the solicitation of proxies and provide related advice and informational support for a services fee and the reimbursement of customary disbursements, which are not expected to exceed $25,000 in the aggregate. We may use severalassign one or more of our regular employees, who will not be specially compensated, to solicit proxies from our stockholders, either personally or by telephone, Internet, facsimile or special delivery letter.
If you need assistance in submitting your proxy over the Internet or completing your proxy card or have questions regarding the annual meeting, please contact Investor Relations at (424) 248-6500 or ir@pumabiotechnology.com or write to: Puma Biotechnology, Inc., 10880 Wilshire Boulevard, Suite 2150, Los Angeles, CA 90024, Attention: Investor Relations.
This proxy statement contains “forward-looking statements” (as defined in the Private Securities Litigation Reform Act of 1995). These statements are based on our current expectations and involve risks and uncertainties, which may cause results to differ materially from those set forth in the statements. The forward-looking statements may include statements regarding actions to be taken by us. We undertake no obligation to publicly update any forward-looking statement, whether as a result of new information, future events or otherwise. Forward-looking statements should be evaluated together with the many uncertainties that affect our business, particularly those mentioned in the risk factors in Item 1A of our Annual Report on Form 10-K for the year ended December 31, 2015 and in our periodic reports on Form 10-Q and our current reports on Form 8-K.
ELECTION OF DIRECTORS
Under our fourth amended and restated bylaws (“Bylaws”), the number of directors shall be fixed from time to time by resolutions of the directors. Our Board has fixed the current size of the Board at five members.eight members, and we have eight current members of the Board.
In evaluating the suitability of individual Board candidates and members, the Nominating and Corporate Governance Committee and Board consider many factors including experience, wisdom, integrity, skills (such as understanding of finance and marketing), educational and professional background, diversity of gender, race, geography, ethnicity, culture, background and perspective, and willingness to devote adequate time to Board duties. The Board has actively sought and appointed qualified female candidates and other diverse directors. For more information concerning the director recruitment and nominating process, please see “Nominating and Corporate Governance Committee” below.
Based upon the recommendation of our Nominating and Corporate Governance Committee, our Board has nominated Alan H. Auerbach, Alessandra Cesano, Allison Dorval, Michael P. Miller, Jay M. Moyes, Adrian M. Senderowicz, Brian Stuglik, and Troy E. Wilson and Frank E. Zavrl for election re-election as directors to the Board. If elected, each director will serve a one-year term expiring at the close of our next annual meeting in 2017,2025, and until such director’s successor is elected and qualified, or until such director’s earlier resignation or removal. Each of Messrs. Auerbach, Miller, Moyes, and ZavrlStuglik, Ms. Dorval, and Drs. Cesano, Senderowicz and Wilson currently serve on our Board. Biographical information onBoard, and each has agreed to serve if re-elected. If any nominee should become unavailable for election prior to the annual meeting (an event that currently is not anticipated by the Board), the proxies will be voted in favor of the election of a substitute nominee or nominees is furnished below under “Director Biographical Information.”proposed by the Board or, alternatively, the authorized number of directors may be reduced accordingly by the Board.
Set forth below is certain information with respect to the nominees.nominees as of the record date. Proxies cannot be voted for a greater number of nominees than the fiveeight nominees set forth below.
Name | Age | Position with the Company | Director Since | |||||||
Alan H. Auerbach | 46 | President, Chief Executive Officer and Chairman of the Board | 2011 | |||||||
Jay M. Moyes(1)(3)(5) | 62 | Director | 2012 | |||||||
Adrian M. Senderowicz(4)(6) | 52 | Director | 2015 | |||||||
Troy E. Wilson(2)(6) | 47 | Director | 2013 | |||||||
Frank E. Zavrl(2)(4) | 46 | Director | 2015 | |||||||
Name | Age | Director Since | Audit Committee | Compensation Committee | Nominating and Corporate Governance Committee | Research and Development Committee | ||||||
Alan H. Auerbach | 54 | 2011 | ||||||||||
Alessandra Cesano, M.D., Ph.D. | 63 | 2022 | M | |||||||||
Allison Dorval | 48 | 2021 | M | |||||||||
Michael P. Miller | 67 | 2018 | M | M | ||||||||
Jay M. Moyes* | 70 | 2012 | C | M | ||||||||
Adrian M. Senderowicz, M.D. | 60 | 2015 | M | C | ||||||||
Brian Stuglik, R.Ph. | 64 | 2020 | C | M | ||||||||
Troy E. Wilson, Ph.D., J.D. | 55 | 2013 | M | C | M |
* | Lead Independent Director |
“C” | Current |
“M” | Current Member |
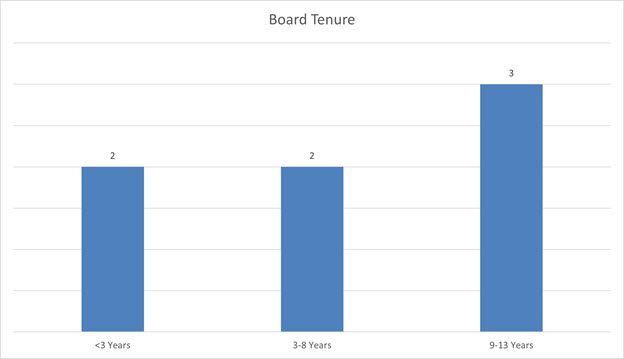
The average tenure of our Board is 7.1 years, comprised of two directors with less than three years of experience, two directors with 3-8 years of experience, and |
Our directors possess a range of diverse skills, backgrounds, experience and viewpoints that we believe are integral to an effective board of directors. The experience matrix below identifies certain of our directors’ experiences, strengths and qualifications by person.
Auerbach | Cesano | Dorval | Miller | Moyes | Senderowicz | Stuglik | Wilson | |
Senior Executive Leadership | ✔ | ✔ | ✔ | ✔ | ✔ | ✔ | ✔ | ✔ |
Other Public Company Board | ✔ | ✔ | ✔ | ✔ | ✔ | ✔ | ✔ | |
Pharmaceutical/Biotechnology | ✔ | ✔ | ✔ | ✔ | ✔ | ✔ | ✔ | ✔ |
Financial/Accounting or Audit | ✔ | ✔ | ✔ | ✔ | ✔ | ✔ |
Commercialization/Sales/Marketing | ✔ | ✔ | ✔ | ✔ |
Research and Product Development | ✔ | ✔ | ✔ | ✔ | ✔ |
Regulatory or Risk Management | ✔ | ✔ |
Advanced Education in Science |
✔ | ✔ | ✔ |
In compliance with NASDAQ’s Board Diversity Rule, the table below provides certain highlights of the composition of our Board members and nominees. These rules require all Nasdaq listed companies to have, or explain why they do not have, at least two diverse directors, including one who self-identifies as female and one who self-identifies as either an under-represented minority or LGBTQ+. Accordingly, we are in compliance with Nasdaq’s Board diversity requirements.
Board Diversity Matrix (As of April 22, 2024) | ||
Total Number of Directors | 8 | |
Female | Male | |
Part I: Gender Identity | ||
2 | 6 | |
Part II: Demographic Background | ||
Hispanic or Latinx | — | 1 |
White (not of Hispanic or Latinx origin) | 2 | 5 |
LGBTQ+ | 1 | |
Director Biographical Information
The following biographical information is furnished with respect to our current directors (including nominees).who are nominees for re-election at the annual meeting.
Alan H. AuerbachAuerbach. . Mr. Auerbach has served as Chairman of our Board and as our President, and Chief Executive Officer and Secretary since October 2011. Prior to October 2011, he served in such capacity at Puma Biotechnology, Inc. (“Puma”), a privately held Delaware corporation and our predecessor, from itsthe Company’s inception in September 2010. Prior to founding Puma,the Company, Mr. Auerbach founded Cougar Biotechnology, Inc. (“Cougar”), a biotechnology company, in May 2003 and served as its Chief Executive Officer, President and a member of its board of directors until July 2009, when Cougar was acquired by Johnson & Johnson. From July 2009 until January 2010, Mr. Auerbach served as the Co-Chairman of the Integration Steering Committee at Cougar (as part of Johnson & Johnson) that provided leadership and oversight for the development and global commercialization of Cougar’s lead drug candidate, abiraterone acetate, for the treatment of advanced prostate cancer. Prior to founding Cougar, from June 1998 to April 2003, Mr. Auerbach was a Vice President, Senior Research Analyst at Wells Fargo Securities, where he was responsible for research coverage of small- and middle-capitalization biotechnology companies, with a focus on companies in the field of oncology. Mr. Auerbach has served as a director of Radius Health, Inc., a public
pharmaceutical company focused on acquiring and developing new therapeutics for the treatment of osteoporosis and other women’s health conditions, sincefrom May 2011 to December 2017 and its predecessor entity from October 2010 to May 2011. Mr. Auerbach received a B.S. in Biomedical Engineering from Boston University and an M.S. in Biomedical Engineering from the University of Southern California. Mr. Auerbach was nominated to serve as a director because of his position as our President and Chief Executive Officer and his significant experience as an executive and research analyst in the biotechnology industry.
Our employment agreement with Mr. Auerbach dated January 19, 2012 provides that Mr. Auerbach will be nominated for election to our Board if the term of his directorship expires during the term of the employment agreement. The original term of his employment agreement was through September 1, 2014, but is subject to successive automatic one-year renewal terms.
AlessandraCesano, M.D., Ph.D. has been a director since July 2022. Dr. Cesano has been the Chief Medical Officer of ESSA Pharma Inc., a pharmaceutical company developing therapies for the treatment of prostate cancer, since July 2019 and Executive Vice President since 2023. Dr. Cesano has also served as a member of the board of directors of Summit Therapeutics Inc., a public biopharmaceutical oncology company, since November 2022, and a member of the board of directors of Zymeworks, a public, clinical-stage biotechnology company, since February 2024. From July 2015 until June 2019, Dr. Cesano was the Chief Medical Officer of NanoString Inc., a biotechnology company that develops translational research tools, where she focused on the development of translational and diagnostic multi-plexed assays for the characterization and measurement of mechanisms of immune response/resistance. Prior to NanoString, Dr. Cesano was Chief Medical Officer at Cleave Biosciences, Inc., a biopharmaceutical company focusing on protein therapies for the treatment of cancer and neurodegenerative diseases, and before that she served as Chief Medical Officer and Chief Operations Officer at Nodality, Inc., where she built and led the Research & Development group, while providing the overall clinical vision for the organization. Between 1998 and 2008, Dr. Cesano held various management positions at Amgen Inc., Biogen Inc. (formerly Biogen Idec) and SmithKline Beecham Pharmaceuticals, where she helped to advance various oncology drugs through late stage development and FDA approvals. Early in her professional career, she spent 12 years conducting research in tumor immunology, including nine years at the Wistar Institute, an NCI Basic Cancer Center connected with the University of Pennsylvania. Dr. Cesano also holds membership in several professional and scientific societies including ASCO, ESMO, ASH, EHA, AACR and the Society of Immunotherapy of Cancer (“SITC”). In the latter, she has served as co-chair in the SITC Industry Committee and of the SITC Biomarker Working Group, and she currently serves as Associate Editor for the Biomarker section of the Journal for ImmunoTherapy of Cancer, co-chair of the Regulatory Committee, and as the elected At-Large Director of the SITC for the 2020-2023 term. Over her career, she has been an author on over 130 publications. Dr. Cesano received an M.D. summa cum laude, a Board Certification in Oncology and a Ph.D. in Tumor Immunology from the University of Turin. Dr. Cesano was selected as a director because of her extensive background in biotechnology research and development and her experience in the life sciences industry.
Allison Dorval. Ms. Dorval has been a director since July 2021. Also in July 2021, Ms. Dorval was appointed to serve on the board of directors of Aerovate Therapeutics, Inc., a public biopharmaceutical company, and has served as the Chief Financial Officer for Verve Therapeutics, Inc. (“Verve”), a public biotechnology company since November 2021. From November 2018 to November 2021, Ms. Dorval served as the Chief Financial Officer of Voyager Therapeutics, Inc. (“Voyager”), a public clinical stage gene therapy company, and as Voyager’s principal financial officer and principal accounting officer. Ms. Dorval joined Voyager as its Vice President of Finance from June 2017 to November 2018. Ms. Dorval is a certified public accountant. Prior to joining Voyager, Ms. Dorval served as Vice President and Controller of Juniper Pharmaceuticals, Inc., a biopharmaceutical company, from August 2016 to June 2017, and as a consultant at Danforth Advisors, a life sciences consultancy focusing on accounting and financial matters, from September 2015 to August 2016. In connection with her role at Danforth, Ms. Dorval served as interim Chief Financial Officer of medical device companies 480 Biomedical, Inc. and Arsenal Medical, Inc. from December 2015 to August 2016. Prior to her time at Danforth, Ms. Dorval served in several roles at Insulet Corporation, a medical device company, from August 2008 to July 2015, including as Chief Financial Officer from November 2014 to May 2015 and as Vice President and Controller from August 2008 to November 2014. Earlier in her career, Ms. Dorval served in various financial and accounting capacities at iBasis, Inc., a telecommunications company; Digitas Inc., an advertising company; and PricewaterhouseCoopers LLP. Ms. Dorval received a B.S. in Business Administration, with a concentration in Accounting, from the University of Vermont and has completed a graduate-level certificate program in Taxation at Bentley University’s McCallum Graduate School of Business. Ms. Dorval was first identified to the Company through an outside search firm, and was subsequently interviewed by Mr. Auerbach and presented to the Nominating and Governance Committee for consideration to be nominated as a director. Ms. Dorval was nominated to serve as a director because of her strong financial background and significant experience in the biopharmaceutical industry.
Michael P. Miller. Mr. Miller has been a director since February 2018. Since 2022, Mr. Miller has served as an advisor to Rigel Pharmaceuticals, a public biotechnology company, Concarlo Therapeutics, a private biotechnology company, and Heathyr, a private diagnostic company. Since January 2022, Mr. Miller has also served on the board of directors for BioXcel, a public biotechnology company. Mr. Miller served as the Executive Vice President U.S. Commercial of Jazz Pharmaceuticals plc, a public biopharmaceutical company from April 2014 until his retirement in September 2020. From April 2010 to January 2014, Mr. Miller was Senior Vice President and Chief Commercial Officer of Vivus, Inc., a public biopharmaceutical company. From 2006 to 2010, Mr. Miller served as Vice President, leading the HER Family Oncology Franchise, of Genentech, Inc., a biotechnology company and wholly owned subsidiary of Roche Holding Ltd. From 2003 to 2005, Mr. Miller served as the Senior Vice President, Chief Commercial Officer of Connetics Corporation, a specialty pharmaceutical company acquired by Stiefel Laboratories, Inc. Previously, from 1997 to 2001, Mr. Miller served as Vice President of the Urology Business Unit of ALZA Corporation, a pharmaceutical company acquired by Johnson & Johnson. Prior to 1997, Mr. Miller served 13 years in various sales and marketing positions at Syntex Corporation, a pharmaceutical company acquired by Roche Holding Ltd. Mr. Miller received a B.S. in Business Administration and Finance from the University of San Francisco and an M.B.A. in Information and Computer Systems from San Francisco State University. Mr. Miller was nominated to serve as a director because of his significant commercialization experience and background in the life sciences industry.
Jay M. Moyes. Mr. Moyes has been a director since April 2012. Mr. Moyes has been a member of the Board and chairmanboard of the audit committeedirectors of Osiris Therapeutics,Biocardia, Inc., a publicly held bio-surgerypublic cardiovascular regenerative medicine company, since May 2006. He has also beenJanuary 2011. Mr. Moyes served as the Chief Financial Officer of Sera Prognostics, Inc., a public commercial-stage biotechnology company focused on improving maternal and neonatal health through innovative biomarker approaches, from March 2020 to June 2023. Mr. Moyes previously served as a member of the board of directors and the chairman of the audit committee for each of Biocardia,Achieve Life Sciences, Inc., a privately held cardiovascularpublic specialty pharmaceutical company, from August 2017 to May 2023; Predictive Technology Group, Inc., a public molecular diagnostics and regenerative medicine company, and Integrated Diagnostics,from February 2019 to December 2019; Osiris Therapeutics, Inc., a privately held molecular diagnosticspublic bio-surgery company, since January 2011from May 2006 until December 2017; and March 2011, respectively. Mr. Moyes was a member of the board of directors of Amedica Corporation, a public orthopedic implant company, from November 2012 to August 2014. He also served as Chief Financial Officer of Amedica from October 2013 to August 2014. From May 2008 through July 2009, Mr. Moyes served as the Chief Financial Officer of XDx (now CareDx), Inc., a privately held molecular diagnostics company. Prior to that, Mr. Moyes served as the Chief Financial Officer of Myriad Genetics, Inc., a publicly heldpublic healthcare diagnostics company, from June 1996 until his retirement in November 2007, and as its Vice President of Finance from July 1993 until July 2005. From 1991 to 1993, Mr. Moyes served as Vice President of Finance and Chief Financial Officer of Genmark, Inc., a privately held genetics company. Mr. Moyes held various positions with the accounting firm of KPMG LLP from 1979 through 1991, most recently as a Senior Manager. He holds an M.B.A. from the University of Utah, a B.A. in economics from Weber State University, and is formerly a Certified Public Accountant. Mr. Moyes also served as a member of the Board of Trustees of the Utah Life Science Association from 1999 through 2006. Mr. Moyes was nominated to serve as a director because of his extensive background in finance and accounting and his experience in the context of the life sciences industry enablesenable him to make significant contributions to the Board.
Adrian M. Senderowicz. Dr. Senderowicz has been a director since August 2015. Dr. Senderowicz has been Senior Advisor of Constellation Pharmaceuticals, Inc., a public clinical-stage biopharmaceutical company focusing on the development of novel tumor-targeted and immuno-oncology therapies, since June 2020 and served as its Senior Vice President and Chief Medical Officer from July 2017 until June 2020. Dr. Senderowicz served as Senior Vice President and Chief Medical Officer of Cerulean Pharma Inc., a public clinical-stage company developing nano-particle conjugates, sincefrom September 2015.2015 until June 2017. Dr. Senderowicz served as the Chief Medical Officer and Senior Vice President, Clinical Development and Regulatory Affairs from August 2014 to February 2015, and Clinical and Regulatory Strategy Officer from February 2015 to AprilMarch 2015 of Ignyta, Inc., a public precision oncology biotechnology company. Prior to joining Ignyta, Dr. Senderowicz was Vice President, Global Regulatory Oncology at Sanofi, a global pharmaceutical company based in France, a position he held from September 2013 to August 2014. Prior to Sanofi, Dr. Senderowicz was Chief Medical Officer and Vice President, Medical Development at Tokai Pharmaceuticals, Inc. from August 2012 to March 2013. From August 2008 to March 2012, Dr. Senderowicz held positions of increasing responsibility, including Senior Medical Director, Oncology Clinical Development, at AstraZeneca.AstraZeneca, a global biopharmaceutical company. Before his tenure at AstraZeneca, Dr. Senderowicz spent almost four years in a variety of leadership positions at the U.S. Food and Drug Administration Division of Oncology Drug Products in the Center for Drug Evaluation and Research. Prior to his work with the FDA,U.S. Food and Drug Administration (“FDA”), Dr. Senderowicz held a variety of clinical and research positions, including Coordinator of the Prostate Cancer Drug Development Clinic and Investigator and Chief, Molecular Therapeutics Unit, with the National Cancer Institute/National Institutes of Health. Dr. Senderowicz holds both an M.D. and an Instructor of Pharmacology degree from the School of Medicine at the Universidad de Buenos Aires in Argentina. Dr. Senderowicz was nominated to serve as a director because of his extensive clinical and regulatory background and his significant experience in the life sciences industry.
Brian Stuglik. Mr. Stuglik has been a director since July 2020. Mr. Stuglik has been a member of the board of directors of Verastem, Inc., a public biopharmaceutical company, since September 2017, where he served as Chief Executive Officer from July 2019 to July 2023. Mr. Stuglik also serves as a member of the board of directors of Oncopeptides AB, a public biotechnology company based in Sweden, since May 2018. Since January 2016, Mr. Stuglik has served as consultant and founder of Proventus Health Solutions, a consulting company for pharmaceutical and biotechnology companies, focusing on U.S. and international pharmaceutical development, product strategy, and commercialization. Prior to founding Proventus Health Solutions, Mr. Stuglik served as Vice President and Chief Marketing Officer for the oncology division of Eli Lilly and Company, a global pharmaceutical company, from 2009 to December 2015. Mr. Stuglik received a B.S. in Pharmacy from Purdue University and holds memberships in the American Society of Clinical Oncology, the American Association of Cancer Research, and the International Association for the Study of Lung Cancer. Mr. Stuglik was nominated to serve as a director because of his significant experience and background in the life sciences industry and, in particular, product strategy and commercialization of pharmaceutical therapies.
Troy E. Wilson. Dr. Wilson has been a director since October 2013. Dr. Wilson has been President and Chief Executive Officer and Chairman of the board of directors of Kura Oncology, Inc., a public clinical-stage biopharmaceutical company that discovers and develops personalized therapeutics for the treatment of solid tumors and blood cancers, since August 2014. Dr. Wilson also serves as a member of the board of directors of Avidity Biosciences, a public biotechnology company, since November 2012. He served as President and Chief Executive Officer of Avidity Biosciences, Inc., a public biopharmaceutical company, from November 2012 to February 2019 and as President and Chief Executive Officer of Wellspring Biosciences, Inc., a private biopharmaceutical company, and its parent company Araxes Pharma LLC, a private biopharmaceutical company, from July 2012 to March 2019. Dr. Wilson served as President and Chief Executive Officer and a member of the board of directors of Kura Oncology,Intellikine, Inc., a public reporting clinical stage biopharmaceutical company discovering and developing personalized therapeutics for the treatment of solid tumors and blood cancers, since August 2014. He has also been the President and Chief Executive Officer and a member of the board of managers of Avidity NanoMedicines LLC, a private biopharmaceutical company, since November 2012 and the President and Chief Executive Officer and a member of the board of
managers of Wellspring Biosciences LLC, a private biopharmaceutical company, since July 2012 and May 2012, respectively. Dr. Wilson served as the President and Chief Executive Officer and a member of the board of directors of Intellikine, a private biopharmaceutical company, from April 2007 to January 2012 and from August 2007 to January 2012, respectively. He has served as a director of Zosano Pharma Corporation, a public clinical stage specialty pharmaceutical company that has developed a proprietary transdermal microneedle patch system to deliver its proprietary formulations of existing drugs through the skin for the treatment of a variety of indications, since June 2014, and as a member of the board of managers of Araxes Pharma LLC, a private biopharmaceutical company, since May 2012. He holds a J.D. from New York University and graduated with a Ph.D. in bioorganic chemistry and a B.A. in biophysics from the University of California, Berkeley. Dr. Wilson was nominated to serve as a director because of his executive leadership experience, his background in finance and accounting and his extensive experience in the life sciences industry.
Frank E. Zavrl. Mr. Zavrl has been a director since September 2015. Mr. Zavrl served as a Partner at Adage Capital Management, L.P. from 2002 to 2011, specializing in biotechnology investments. Prior to joining Adage Capital, Mr. Zavrl was a Portfolio Manager from 1999 to 2002 at Merlin Biomed, a healthcare investment group. From 1998 to 1999, Mr. Zavrl was an analyst at Scudder Kemper Investments Inc., focusing on biotechnology investments. Mr. Zavrl received a B.S. in Biochemistry from the University of California, Berkeley and an M.B.A. from the Tuck School of Business at Dartmouth College. Mr. Zavrl was nominated as a director because of his significant experience and background in the biotechnology investments field.
OUR BOARD UNANIMOUSLY RECOMMENDS A VOTE “FOR” THE ELECTION OF EACH OF THE FIVE DIRECTOR NOMINEES.
Set forth below is information regarding each of our executive officers as of the date of this proxy statement.
|
| |||||
| ||||||
| ||||||
| ||||||
| ||||||
| ||||||
Alan H. Auerbach. See “Director Biographical Information” above.
Charles R. Eyler. Mr. Eyler has served as our Senior Vice President, Finance and Administration and Treasurer since October 2011. Prior to October 2011, he served in such capacity at Puma beginning in September 2011. Prior to joining Puma, Mr. Eyler served as Senior Vice President of Finance at Cougar until July 2009, when Cougar was acquired by Johnson & Johnson. He also served as Treasurer of Cougar from April 2006 to July 2009. From July 2009 until March 2010, Mr. Eyler served on the Integration Steering Committee at Cougar (as part of Johnson & Johnson) and oversaw the integration of Cougar’s finance and IT functions with those of Johnson & Johnson. From April 2010 until September 2011, Mr. Eyler explored various entrepreneurial and other opportunities. Prior to joining Cougar, Mr. Eyler served as Chief Financial Officer and Chief Operating Officer of Hayes Medical Inc. from March 1999 to January 2004. Mr. Eyler received his B.S. from Drexel University and his M.B.A. from Saint Francis College.
Richard P. Bryce, MBChB, MRCGP and MFPM. Dr. Bryce has served as our Senior Vice President, Clinical Research and Development since June 2012. Dr. Bryce previously served as Senior Medical Director for Onyx Pharmaceuticals, a biopharmaceutical company, from September 2008 to June 2012, where he oversaw the Phase III clinical trial program of carfilzomib for the treatment of multiple myeloma and the Phase II clinical trial program of sorafenib for the treatment of breast and colorectal cancers. From August 2007 to August 2008, Dr. Bryce served as Senior Medical Director for ICON Clinical Research, a clinical research organization, where he was responsible for developing and evaluating oncology protocols, medical monitoring, and overseeing drug safety management activities in connection with the clinical trials of oncology drugs. From May 2005 until July 2007, he served as Executive Vice President of Medical Affairs at Ergomed Clinical Research, a clinical research organization, where he worked to establish the company’s U.S. operations, had overall responsibility for the global Phase I unit activities, drug safety, medical writing and regulatory affairs, and oversaw the company’s provision of consulting services to various oncology-focused biotechnology companies. From April 2003 to May 2005, Dr. Bryce served as International Medical Leader at Roche, where he oversaw the global Phase IV clinical trial program of Xeloda® (capecitabine) for the treatment of breast cancer. Dr. Bryce holds a BSc in Medical Sciences and his primary medical degree (MBChB) from the University of Edinburgh, Scotland. He also holds post-graduate diplomas in Obstetrics and Gynaecology from the Royal College of Obstetricians and Gynaecologists of London and in Child Health and Pharmaceutical Medicine from the Royal College of Physicians of the United Kingdom. He is a member of the Royal College of General Practitioners and the Royal College of Physicians (Faculty of Pharmaceutical Medicine) of the United Kingdom. He is also a member of the American Society of Clinical Oncology, the American Society of Hematology and the European Society of Medical Oncology.
Steven Lo. Mr. Lo has served as our Chief Commercial Officer since September 2015. Prior to joining the Company, Mr. Lo held a number of positions at Corcept Therapeutics Incorporated from September 2010 to September 2015, including Senior Vice President & Head of Oncology, Senior Vice President & Chief Commercial Officer and Vice President & Head of Commercial Operations. Prior to Corcept, Mr. Lo was with Genentech, Inc. from December 1997 to September 2010. At Genentech, Mr. Lo held a number of positions, including Senior Director, Oncology Marketing, where he prepared and led the first U.S. launch of Herceptin® in adjuvant HER2-positive breast cancer and also worked with Genentech’s then ex-U.S. marketing partner, Roche, to develop the global adjuvant launch strategy for Herceptin® in adjuvant HER2-positive breast cancer. Mr. Lo received a B.S. in Microbiology from the University of California, Davis and a Master of Health Administration from the University of Southern California.
Richard B. Phillips, Ph.D. Dr. Phillips has served as our Interim Head of Regulatory Affairs, Quality Assurance and Pharmacovigilance since April 2016. Dr. Phillips previously served in a similar capacity at the Company as Senior Vice President, Regulatory Affairs, Quality Assurance and Pharmacovigilance from November 2011 to November 2014. From March 2010 to October 2011, he worked as a consultant with pharmaceutical and biotech companies in the area of regulatory affairs. From January 2007 to July 2009, Dr. Phillips served as Senior Vice President of Regulatory Affairs and Quality Assurance at Cougar Biotechnology, Inc., and following the acquisition of Cougar by Johnson & Johnson, from July 2009 until March 2010, he oversaw the integration of Cougar’s regulatory affairs and quality assurance function with Johnson & Johnson. From September 2005 to January 2007, Dr. Phillips was employed by Amgen Inc., where he was the Director of Regulatory Affairs and Global Regulatory Leader for Vectibix (panitumumab), which received FDA approval in 2006 for the treatment of metastatic colorectal cancer. Dr. Phillips has also held regulatory affairs management positions with Chugai Pharma USA, Pfizer Inc. (Parke-Davis), Johnson & Johnson (Janssen, L.P.), Novartis A.G., G.D. Searle (Pfizer) and Structural GenomiX. Dr. Phillips received a B.S. from the University of California, Irvine and a Ph.D. from the University of California, Berkeley.
None of our directors, nominees or executive officers is related by blood, marriage or adoption to any other director, nominee or executive officer. In addition, exceptExcept as indicated herein with respect to Mr. Auerbach, no arrangements or understandings exist between any director or person nominated for election as a director and any other person pursuant to whom such person is to be selected as a director or nominee for election as a director.
OUR BOARD UNANIMOUSLY RECOMMENDS A VOTE “FOR” THE ELECTION OF EACH OF THE EIGHT DIRECTOR NOMINEES.
Board Leadership StructureIndependence
Our Board has determined that seven of our eight directors are independent, and that each of the members of our Audit Committee, Compensation Committee, Nominating and Corporate Governance Committee and Research and Development Committee is independent. In making these determinations, our Board has made a subjective determination as to each independent director that no relationships exist, which, in the opinion of our Board, would interfere with the exercise of independent judgment in carrying out the responsibilities of a director.
On an annual basis, our Board undertakes a review of its composition, the composition of its committees and the independence of each director. For the purpose of its independence determinations, the Board employs the standards for independence set forth in the listing requirements and rules of The NASDAQ Stock Market LLC (“NASDAQ”), in the rules and standards established by the U.S. Securities and Exchange Commission (the “SEC”) and in our corporate governance guidelines. Our corporate governance guidelines are available on our corporate website at https://www.pumabiotechnology.com/about_governance.html.
Based upon information requested from and provided by each of our directors concerning his or her background, employment and affiliations, including family relationships, and on such other due consideration and diligence as it deems appropriate, our Board has determined that Messrs. Miller, Moyes, and Stuglik, Ms. Dorval and Drs. Cesano, Senderowicz and Wilson, or seven of our eight current directors, are “independent” under the applicable rules and standards established by NASDAQ and the SEC, as well as under the additional standards set forth in our corporate governance guidelines. In making this determination, our Board considered the current and prior relationships that each non-employee director has with us and all other facts and circumstances our Board deemed relevant in determining their independence, including the beneficial ownership of our capital stock by each non-employee director. Our Board has determined that Mr. Auerbach is not independent due to his role as our President and Chief Executive Officer.
Alan H. Auerbach currently serves as our Chairman and Chief Executive Officer. We have no policy requiring the combination or separation of the Chief Executive Officer and Chairman roles and our governing documents do not mandate a particular structure. At present, we have determined that this leadership structure of having a combined Chairman of the Board and Principal Executive Officer is appropriate due to the size and operations and resources of our company. Our Board believes that having these roles combined helps promote efficient and centralized decision-making, focuses the Board’s discussions and facilitates the presentation of the Company’s strategy with a unified voice.
Our Board acknowledges that no single leadership model is right for all companies at all times. As such, our Board periodically reviews its leadership structure and may, depending on the circumstances, including our size, resources and operations, choose a different leadership structure in the future.
Our Board is involved in the general oversight of risks that could affect our business. Our Board satisfies this responsibility through reports by each committee chair regarding the committee’s considerations and actions, as well as through regular reports directly from officers responsible for oversight of particular risks within the Company. Further, our Board oversees risks through the establishment of policies and procedures that are designed to guide daily operations in a manner consistent with applicable laws, regulations and risks acceptable to our Company. The role of the Board’s committees in overseeing many of the risks associated with our business includes the following:
• | The Audit Committee oversees and reviews with management our assessment and management process of material financial reporting and accounting risks, including review of internal controls over financial reporting, policies, practices and risks related to cybersecurity and disclosure controls and procedures, as well as reviews and approves any related party transactions. |
• | The Compensation Committee oversees and assesses whether any of our compensation plans, policies, and programs are reasonably likely to have a material adverse effect on us. |
• | The Nominating and Corporate Governance Committee oversees and reviews with management risks relating to governance, environmental and social responsibility matters, including succession planning. |
• | The Research and Development Committee oversees and reviews with management evaluation of our product pipeline and pre-trial and clinical development risks. |
The Board’s, including the Research and Development Committee, 2023 review included an assessment of the risks related to effects of the closure of clinical trials of NERLYNX with respect to our commercialization of NERLYNX. The Research and Development Committee also assessed the clinical development risks associated with our acquisition of worldwide research and development, and commercial, rights to alisertib.
Board IndependenceLeadership Structure
Under the listing requirements and rules of the New York Stock Exchange (the “NYSE”), independent directors must comprise a majority of a listed company’s board of directors. In addition, NYSE rules require that, subject to specified exceptions, each member of a listed company’s audit, compensation and nominating and corporate governance committees be independent. Audit committee members must also satisfy the independence criteria set forth in Rule 10A-3 under the Securities Exchange Act of 1934, as amended (the “Exchange Act”), and compensation committee members must satisfy heightened independence criteria set forth in NYSE rules. Under NYSE rules, a director will only qualify as an “independent director” if the company’s board of directors affirmatively determines that the director has no material relationship with the company, either directly or indirectly, that would interfere with the exercise of independent judgment in carrying out the responsibilities of a director.
Our Board has undertaken a review of its composition, the composition of its committees and the independence of each director. Based upon information requested from and provided by each of our directors concerning his background, employment and affiliations, including family relationships, and on such other due consideration and diligence as it deems appropriate, our Board has determined that each of Messrs. Moyes and Zavrl and Drs. Senderowicz and Wilson is “independent” under the applicable rules and standards established by the U.S. Securities and Exchange Commission (the “SEC”) and the NYSE. In making this determination, our Board considered the current and prior relationships that each non-employee director has with us and all other facts and circumstances our Board deemed relevant in determining their independence, including the beneficial ownership of our capital stock by each non-employee director. Our Board has determined that Mr.Alan H. Auerbach is not independent due to his rolecurrently serves as our PresidentChairman and Chief Executive Officer. We have no policy requiring the combination or separation of the Chief Executive Officer and Chairman roles and our governing documents do not mandate a particular structure. At present, we have determined that this leadership structure of having a combined Chairman of the Board and Chief Executive Officer is appropriate due to the size and operations and resources of our Company. Our Board believes that having these roles combined helps promote efficient and centralized decision-making, focuses the Board’s discussions and facilitates the presentation of the Company’s strategy with a unified voice. Our Board also believes in the value and importance of a strong Lead Independent Director with clearly delineated responsibilities, including to preside over Board meetings at which the Chief Executive Officer and Chairman is not present (including any executive sessions of the independent directors), approve Board meeting schedules and agendas and act as liaison between the independent directors and the Chief Executive Officer and Chairman. Mr. Moyes serves as the Lead Independent Director.
Our Board acknowledges that no single leadership model is right for all companies at all times. As such, our Board periodically reviews its leadership structure and may, depending on the circumstances, including our size, resources and operations, choose a different leadership structure in the future.
During the fiscal year ended December 31, 2015,2023, our Board held six meetings. Allheld five meetings. All directors attended at leastleast 75% or moreor more of the aggregate number of meetings of the Board and board committees on which they served. We do not have a formal policy relatingAs part of our director education program, each director is strongly encouraged to director attendance at annual meetings. Mr. Auerbach attended our 2015attend each annual meeting of stockholders held on June 9, 2015.
During the fiscal year ended December 31, 2015,2023, the non-executivenon-employee directors met in executive session of the Board on fourfive occasions, and the members of the Audit Committee met in executive session on fivefour occasions, the members of the Compensation Committee met in executive session on four occasions and acted by unanimous written consent on ten occasions, and the members of the Nominating and Corporate Governance Committee met in executive session on two occasions. The policy of our Board is to hold at least four executive sessions of thein connection with each regularly scheduled Board annuallymeeting and executive sessions of committees when needed. Jay M.As our Lead Independent Director, Mr. Moyes presidespresided over the regularly scheduled executive sessions of the non-management directors.Board.
We have established an Audit Committee, a Compensation Committee, a Nominating and Corporate Governance Committee, an Equity Incentive Committee and a Research and a Stock OptionDevelopment Committee. The composition and responsibilities of each committee are determined by the Board and are described below. To view the charters of the Audit Committee, Compensation Committee, and Nominating and Corporate Governance Committee and Research and Development Committee, please visit the corporate governance section of our website athttps://www.pumabiotechnology.com/about_governance.html. In addition, the charters for these committees are available in print to any stockholder who requests a copy. Please direct all requests to our Corporate Secretary, Puma Biotechnology, Inc., 10880 Wilshire Boulevard, Suite 2150, Los Angeles, CA 90024.
Audit Committee
Our Audit Committee provides oversight over each of our accounting and financial reporting processes, our internal control function, the auditaudits of our consolidated financial statements and our internal control function.over financial reporting. Among other matters, the Audit Committee assists our Board in oversight of the independent registered public accounting firm qualifications, independence and performance; is responsible for the engagement, retention and compensation of the independent auditors; reviews the scope of the annual audit; reviews and discusses with management and the independent registered public accounting firm the results of the annual audit and the review of our quarterly consolidated financial statements, including the disclosures in our annual and quarterly reports filed with the SEC; reviews our risk assessment and risk management processes; establishes procedures for receiving, retaining and investigating complaints received by us regarding accounting, internal accounting controls or audit matters; approves audit and permissible non-audit services provided by our independent registered public accounting firm; and reviews and approves related personparty transactions under Item 404 of Regulation S-K.S-K and our Related Party Transactions Policy and Procedures.
The members of our Audit Committee are Messrs.Mr. Moyes, and ZavrlMs. Dorval and Dr. Wilson with Mr. Moyes serving as the chair of the committee. The Board has determined that each of Messrs.Mr. Moyes, and ZavrlMs. Dorval and Dr. Wilson are independent directors as defined under the applicable rules and regulations of the SEC and NASDAQ and the NYSE.requirements in the Audit Committee’s charter and our corporate governance guidelines. The Board has determined that all members of our Audit Committee meet the requirements for financial literacyservice as an audit committee member under the applicable rules and regulations of the SECNASDAQ, and the NYSE. Our Board has determined that Mr. Moyes, Ms. Dorval and Dr. Wilson are audit committee financial experts as defined under the applicable rules of the SEC and have the requisite financial sophistication as defined under the applicable rules and regulations of the NYSE.SEC. The Audit Committee met ninefour times during the fiscal year ended December 31, 2015.2023. The Audit Committee meets in executive session for a portion of each regular meeting.
Our Board considers cybersecurity risk as part of its risk oversight function and has delegated to the Audit Committee oversight of cybersecurity and other information technology risks. The Audit Committee oversees management’s implementation of our cybersecurity risk management program. The Audit Committee receives periodic reports from management on our cybersecurity risks. In addition, management updates the Audit Committee, as necessary, regarding any material cybersecurity incidents, as well as any incidents with lesser impact potential.
The Audit Committee reports to the Board regarding its activities, including those related to cybersecurity. The Board also receives briefings from management on our cyber risk management program. Members of the Board receive presentations on cybersecurity topics from internal security staff or external experts as part of the Board’s continuing education on topics that impact public companies. Our management team, including our Chief Financial Officer, Senior Director of Information Technology and Senior Vice President, Quality Assurance, is responsible for assessing and managing our material risks from cybersecurity threats. The team has primary responsibility for our overall cybersecurity risk management program and supervises both our internal technology and quality assurance compliance personnel and our retained external cybersecurity consultants. Our management team has experience in risk management as well as in the technology and finance industries, equipping them to oversee cybersecurity risks effectively.
Compensation Committee
Our
The Compensation Committee adopts and administers the compensation policies, plans and benefit programs for our executive officers and all other members of our executive team.officers. In addition, among other things, ourthe Compensation Committee annually evaluates the performance and compensation of our Chief Executive Officer and, in consultation with our Chief Executive Officer, our other executive officers. Based on such evaluation, the Compensation Committee determines and approves all of the compensation of the Chief Executive Officer and other executive officers. The Chief Executive Officer is not permitted to be present during any Compensation Committee final deliberations or voting concerning the compensation of any executive officer, including the Chief Executive Officer. OurThe Compensation Committee also administers the 2011 Plan and the Puma
Biotechnology, Inc. 20112017 Employment Inducement Incentive Award Plan (the “Plan”“Inducement Plan” and, together with the 2011 Plan, the “Plans”). Additionally, the Compensation Committee annuallyperiodically reviews and makes recommendations to our Board regarding the compensation and benefits of our non-managementnon-employee directors.
The Compensation Committee is permitted to delegate any or all of its responsibilities to a subcommittee of the Compensation Committee, but only to the extent consistent with our Certificate of Incorporation, Bylaws, Section 162(m) of the Internal Revenue Code of 1986, as amended (the “Code”), the rules and listing standards of the NYSE,NASDAQ, and other applicable law. The Compensation Committee has delegated certain of its authority under the Plan to the Stock Option Committee. See “Stock Option Committee.”
The Compensation Committee has the sole discretion to retain or obtain the advice of compensation advisers,advisors, including compensation consultants, legal counsel or other advisersadvisors in order to assist the Compensation Committee or any of its subcommittees in carrying out its responsibilities. The Compensation Committee is also responsible for the appointment, determination of the compensation and oversight of the work of so retained compensation advisersadvisors and the determination of the independence of each compensation adviseradvisor prior to selecting or receiving advice from any such compensation adviseradvisor and on at least an annual basis thereafter. The Company provides for appropriate funding for payment or reasonable compensation to any compensation adviseradvisor to the Compensation Committee.
The
In 2023, the Compensation Committee has engaged Radford (the “consultant” or “Radford”continued to engage Compensia, Inc. (“Compensia”), an independent compensation consultant, to advise the Compensation Committee, on an ongoingand the advice and comprehensive report received from Compensia in 2022 served as the basis as an independentfor the Compensation Committee’s compensation consultant. The consultant reportsrelated decisions for 2023. Compensia reported directly to the Compensation Committee. While conducting assignments, the consultant interacts with our management when appropriate. Specifically, our Senior Vice President, FinanceCommittee and Administration and Treasurer and our senior finance and human resources personnel interact with the consultant from time to time to provide relevant company and executive compensation data. In addition, the consultant may seek feedback from the Chairman of theattended Compensation Committee other members of our Board or the Chief Executive Officer regarding its work prior to presenting study results or recommendations to the Compensation Committee.meetings in 2023 following invitations. The Compensation Committee determines when to hire, terminate or replace the consultant, andcompensation consultants, the projects to be performed by the consultant. During 2015,any consultant and whether the consultant at the requestis invited to attend meetings of the Compensation Committee. In early 2023, the Compensation Committee performedobtained a review ofnew detailed compensation analysis which served as the competitiveness of ourbasis for the Compensation Committee’s compensation programsrelated decisions for certain of our senior management.2023. The Compensation Committee may engage the consultantCompensia to conduct additional comprehensive reviews of our senior management compensation programs in the future. In addition, in the future, the consultant, when invited, may attend meetings of the Compensation Committee.
The Compensation Committee reviews the independence of its compensation consultants and other advisors. In performing its analysis, the Compensation Committee considers the factors set forth in applicable SEC rules and NASDAQ listing requirements and the NYSE listing requirements.requirements in the Compensation Committee’s charter and our corporate governance guidelines. After review and consultation with Radford,Compensia, the Compensation Committee has determined Radfordthat Compensia is independent and that there is no conflict of interest resulting from retaining RadfordCompensia currently nor was any conflict raisedor during the year ended December 31, 2015.2023. In reaching these conclusions, the Compensation Committee considered the factors set forth in the Securities Exchange Act of 1934, as amended (the “Exchange Act”) Rule 10C-1 and NASDAQ listing standards.
The members of ourthe Compensation Committee are Messrs. Miller, Moyes and Zavrl and Dr. Senderowicz,Stuglik with Mr. MoyesMiller serving as chair of the committee until October 1, 2023, and Mr. Stuglik serving as the chair of the committee.committee as of October 1, 2023. The Board has determined that each of the members of ourthe Compensation Committee areis independent under the applicable rules and regulationslisting standards of NASDAQ and is independent under the SECrequirements in the Compensation Committee’s charter and the NYSE, are “outside directors” for purposes of Section 162(m) of the Codeour corporate governance guidelines and are non-employee directors for purposes of Section 16 of the Exchange Act. The Compensation Committee met four times during the fiscal year ended December 31, 2015.2023 and acted by unanimous written consent on ten occasions. Additionally, matters delegated to the Compensation Committee were discussed during meetings of the Board, including in executive session with only independent members of the Board present.
Nominating and Corporate Governance Committee
Our Nominating and Corporate Governance Committee is responsible for, among other things, making recommendations regarding corporate governance, the composition of our Board, identification, evaluation and nomination of director candidates and the structure and composition of committees of our Board. In addition, our Nominating and Corporate Governance Committee oversees our corporate governance guidelines, makes
recommendations regarding our committee charters, oversees compliance with our code of business conduct and ethics, contributes to succession planning, reviews actual and potential conflicts of interest of our directors and officers other than related personparty transactions reviewed by the Audit Committee and oversees the self-evaluation process of our Board. Our Nominating and Corporate Governance Committee also is responsible for making recommendations regarding non-employee director compensation to the full Board.Board and oversees and reviews with management risks relating to governance, environmental and social responsibility matters.
The process followed by our Nominating and Corporate Governance Committee to identify and evaluate director candidates includes meetingsmeeting from time to time to evaluate biographical information and background material relating to potential candidates and, as deemed advisable, interviews of selected candidates by our Chief Executive Officer, members of the Nominating and Corporate Governance Committee andor other members of the Board. It is the policy of the Nominating and Corporate Governance Committee that when it establishes an initial list of potential candidates for consideration as a new candidate to the Board, the committee shall include in such list, but shall not be limited to, one or more qualified women and minority candidates. The Nominating and Corporate Governance Committee may seek the assistance of an outside search firm in identifying and evaluating potential candidates from time to time. Dr. Cesano was first identified to the Company through an outside search firm, which was hired to identify potential suitable Board candidates for consideration.
In evaluating the suitability of individual candidates (both new candidates and current Board members) to recommend to the Board for nomination (and, in the case of vacancies, appointment), the Nominating and Corporate Governance Committee appliesand the Board reviews the candidate in the context of the criteria attached to its charter. These criteria include (i) personal and professional integrity, ethics and values; (ii) experience in corporate management, such as serving as an officer or former officer of a publicly held company, and a general understanding of marketing, finance and other elements relevant to the success of a publicly held company in today’s business environment; (iii) experience in our industry and with relevant social policy concerns; (iv) experience as a board member of another publicly held company; (v) academic expertise in an area of our operations; and (vi) practical and mature business judgment, including ability to make independent analytical inquiries.inquiries; and (vii) diversity of gender, race, geography, ethnicity, culture, background, viewpoints and perspective. Each individual is evaluated in the context of the Board as a whole, with the objective of assembling a groupboard of directors that can best perpetuate the success of the business and represent stockholder interests through the exercise of sound judgment using his or her diversity of experience in these various areas. While theThe Nominating and Corporate Governance Committee considers diversityis mindful of the policies regarding board service of certain investors and varietyproxy advisory firms, which were developed due to concerns that “overboarded” directors face excessive time commitments and challenges in fulfilling their duties. Our corporate governance guidelines prohibit directors from serving on the boards of experiences and viewpoints to be important factors, it does not believe that a director nominee should be chosen solely or mainly becausemore than four public companies, including the Company, without the Board’s consent. All of race, color, gender, national origin or sexual identity or orientation. Thus, although diversity may be a considerationour nominees are in the Nominating and Corporate Governance Committee’s process, it does not have a formal policy regarding the consideration of diversity in identifying director nominees.
Additionally, thecompliance with this requirement. The Nominating and Corporate Governance Committee does not have a formal policy with regard toreviews and considers the contributions and performance of each individual, as well as consideration of any director candidates recommended by stockholders. Because ofwhether a director’s service on other boards will enhance, rather than interfere with, their service on our Board. In making its nominee recommendations for the size of the Board, theAnnual Meeting, our Nominating and Corporate Governance Committee addressesnoted the need to retain membersstrong attendance, preparedness and fill vacancies after discussion among current members. Accordingly, theengagement at Board and committee meetings, dedication through committee membership and compliance with our governance guidelines for each nominee. The Nominating and Corporate Governance Committee has determined thatalso values their input resulting from experience as a formal policy for consideringpublic company director candidates recommended by stockholders is not necessary.and extensive expertise in the biopharmaceutical and biotechnology industries, and believes the nominees’ service on other boards enhances their contributions and the strength of our Board.
The Nominating and Corporate Governance Committee will however, consider director candidates recommended by stockholders even though it has no requirement to do so. The Nominating and Corporate Governance Committeeany stockholder who holds 5% or more of our common stock. Such candidate will consider such candidatesbe reviewed by the committee on the same basis as it considers all other candidates. A stockholder wishing to submit a director nomination should send a letter to the Board of Directors, c/o Corporate Secretary, Puma Biotechnology, Inc., 10880 Wilshire Boulevard, Suite 2150, Los Angeles, CA 90024. The mailing envelope must contain a clear notation indicating that the enclosed letter is a “Director Nominee Recommendation.” A stockholder that wishes to nominaterecommend a director candidate should submit complete information as to the identity and qualifications of the director candidate to the Nominating and Corporate Governance Committee, including all information that would be required to be disclosed about that person in a proxy statement relating to the election of directors. In making recommendations, stockholders should be mindful of the discussion of the minimum qualificationscriteria set forth above. Satisfaction of such minimum qualification standardscriteria does not imply that the Nominating and Corporate Governance Committee necessarily will nominate the person so recommended by a stockholder. With respect to deadlines and other matters relating to stockholder nominations of director candidates, see “Stockholder Proposals and Nominations.”
The members of our Nominating and Corporate Governance Committee are Mr. MoyesMiller and Drs. Senderowicz and Wilson, with Mr. MoyesDr. Wilson serving as the chair of the committee. The Board has determined that each of the members of our Nominating and Corporate Governance Committee areis independent under the
applicable rules and regulations of the SEC and listing standards of NASDAQ and the NYSE.requirements in the Nominating and Corporate Governance Committee’s charter and our corporate governance guidelines. The Nominating and Corporate Governance Committee met three timeson two occasions during the fiscal year ended December 31, 2015.2023.
Stock OptionResearch and Development Committee
Our Research and Development Committee oversees our product pipeline and research and development efforts, including oversight and evaluation of our clinical trials and clinical development risk, as well as reviewing and pre-approving all material public disclosures related to our product pipeline and research and development efforts. As part of its oversight responsibilities, the committee also meets at least quarterly with the Chief Scientific Officer to review the progress of the product pipeline and the progress and results of pre-clinical studies and clinical trials. The Research and Development Committee is comprised of Drs. Senderowicz, Wilson and Cesano and Mr. Stuglik, with Dr. Senderowicz serving as the chair of the committee. The Board has determined that each of the members of our Research and Development Committee is independent under the requirements in the Research and Development Committee’s charter and our corporate governance guidelines. The Board has determined that Drs. Senderowicz, Wilson and Cesano and Mr. Stuglik meet the requirements for scientific and/or medical expertise under the Research and Development Committee’s charter. The Research and Development Committee’s discussions included an assessment of the risks related to effects of the closure of clinical trials of NERLYNX with respect to our commercialization of NERLYNX as well as the assessment of the clinical development risks associated with the acquisition of worldwide research and development, and commercial, rights to alisertib.
Equity Incentive Committee
Our Stock OptionEquity Incentive Committee consists of Mr. Auerbach, a director and our President and Chief Executive Officer, serving as its sole member. The Board delegated to the Stock OptionEquity Incentive Committee the authority to grant stock options and restricted stock units (“RSUs”) to non-executive employees, subject to the following conditions:
the maximum aggregate number of shares of common stock underlying options granted pursuant to this authority is 100,000 per individual, subject to adjustment by the Board; and
• | the maximum aggregate number of shares of common stock underlying RSUs granted pursuant to this authority is 100,000 per individual, subject to adjustment by the Board; |
• | the maximum aggregate number of shares of common stock underlying stock options granted pursuant to this authority is 100,000 per individual, subject to adjustment by the Board; and |
the stock options must have an exercise price equal to the closing price of our common stock on the grant date and have a term not longer than ten years.
• | the stock options must have an exercise price equal to the closing price of our common stock on the grant date and have a term not longer than 10 years. |
Pursuant to this delegation of authority, for fiscal year 2015,2023, the Stock Option CommitteeEquity Incentive Committee granted 2,606,183 stock options.an aggregate of 776,276 RSUs.
We are not aware of any material proceedings in which any of our directors, executive officers or affiliates, any owner of record or beneficial owner of more than 5% of our common stock, or any associate of any such director, officer, affiliate or security holder is a party adverse to us or any of our subsidiaries or has a material interest adverse to us.
Code of Business Conduct and Ethics
We have adopted a code of business conduct and ethics that establishes the standards of ethical conduct applicable to all directors, officers and employees of our company.Company. Our code of business conduct and ethics addresses, among other things, conflicts of interest, compliance with disclosure controls and procedures and internal control over financial reporting, corporate opportunities and confidentiality requirements. Our code of business conduct and ethics is available on our corporate website athttps://www.pumabiotechnology.com/about_governance.html.We intend to disclose any future amendments to certain provisions of our code of business conduct and ethics, or waivers of provisions required to be disclosed under the rules of the SEC, at the same location on our website identified in the preceding sentence.
Corporate Governance Guidelines
We have adopted corporate governance guidelines, which, among other things, highlight that the Board should consider diversity of gender, race, geography, ethnicity, culture, background, and perspective in evaluating the suitability of individual members of the Board. Our corporate governance guidelines are available on the Company’sour corporate website athttps://www.pumabiotechnology.com/about_governance.html.
Stockholders and other interested parties may send communications to the Company’s Board, including any individual director, any non-managementnon-employee director or the directors as a group, by mailing such communications to Puma Biotechnology, Inc., 10880 Wilshire Boulevard, Suite 2150, Los Angeles, CA 90024, Attention: Corporate Secretary. Such correspondence shall be addressed to the Board, any individual director or any non-managementnon-employee director by either name or title.
All communications received as set forth in the preceding paragraph will be opened by the Company’s Corporate Secretary for the sole purpose of determining whether the contents represent a message to the Company’s directors. Any contents that are not in the nature of advertising, promotions of a product or service or patently offensive material will be forwarded promptly to the addressee. In the case of communications to the Board, any individual director or any non-managementnon-employee director, the Company’s Corporate Secretary will make sufficient copies of the contents to send to each director to which the envelope is addressed.
Pledging and Hedging Prohibited
We maintain an Insider Trading Compliance Policy that prohibits our officers, directors and employees from pledging Company stock as collateral to secure loans and from engaging in hedging transactions, including zero-cost collars and forward sale contracts. It further prohibits margin purchases of the Company’s stock, short sales of the Company’s stock, and any transactions in puts, calls or other derivative securities involving the Company’s stock.
Environmental, Social and Governance
ESG Governance
Our Nominating and Corporate Governance Committee provides oversight and guidance regarding ESG. This committee periodically discusses ESG topics and interacts with executive leadership on ESG topics. Executive leadership has established a cross-functional ESG Working Group to provide operational guidance and direction regarding implementation of ESG at the Company. The ESG Working Group includes:
| • | Chief Financial Officer |
| • | Chief Accounting Officer |
| • | Senior Vice President, Manufacturing |
| • | Chief Commercial Officer |
| • | Senior Vice President, Quality Assurance |
| • | Vice President, Human Resources |
| • | Senior Director, Investor Relations |
Identifying High-Priority ESG Topics
To identify those ESG topics that are the highest priority for us to focus on, internal and external perspectives on ESG were evaluated. Through a process that is often labeled a “materiality assessment,” the perspectives of internal and external stakeholders were assessed, and we considered the highest priority ESG topics to be those that are most important to both internal and external stakeholders. Internal stakeholder perspectives were provided by the members of the ESG Working Group. External stakeholder perspectives were determined through sources that document the positions of key external stakeholders on ESG issues for the biopharmaceutical industry. Those sources include:
| • | Biopharma Investor ESG Communications Guidance published by Biopharma Sustainability Roundtable |
| • | Sustainability Accounting Standards Board Biopharmaceuticals Standard |
| • | Key ESG ratings and the topics most heavily weighted in those ratings |
| • | Peer company reporting |
The ESG topics identified as being of significant importance to both internal and external stakeholders are:
| • | Access to Medicine/Healthcare |
| • | Business Ethics |
| • | Research and Development (with particular emphasis on Clinical Trial Practices) |
| • | ESG Governance |
| • | Product Quality and Patient Safety |
| • | Pharmaceuticals in the Environment |
It should be noted that these are not the only ESG topics in which we are taking action and implementing programs; rather, these are the topics that are deemed most important and worthy of external reporting at this time. In addition, this list will be reviewed and updated as warranted.
Business Ethics and Compliance
Our Board provides oversight of our compliance program and adherence to high standards of business ethics. The chair of the Audit Committee serves as the primary liaison between the Board and executive leadership on matters of ethics and compliance. Operational guidance and direction for compliance and business ethics are provided by the Compliance Committee, which is chaired by the Ethics and Compliance Officer. Members of the Compliance Committee include the senior leads of the following functions:
| • | Quality Assurance |
| • | Pharmacovigilance |
| • | Human Resources |
| • | Legal |
| • | Commercial |
| • | Regulatory Affairs |
| • | Finance |
| • | Clinical Research and Development |
The Compliance Committee meets on at least a quarterly basis. Amongst other duties, the Compliance Committee ensures that we have devoted sufficient resources to maintain compliance with all applicable regulations and high ethical standards. In particular, the Compliance Committee is charged with ensuring that our compliance program is in alignment with the Office of the Inspector General (OIG) Compliance Program Guidance for Pharmaceutical Manufacturers. In addition, members of the Compliance Committee serve as Compliance Ambassadors within their respective departments to ensure collaboration, awareness and training are maintained.
As part of our overall commitment to operating ethically, we have committed to a “speak-up culture,” which is codified through our Compliance Reporting and Investigations Policy. This policy includes the commitment to maintaining a 24/7 reporting hotline that allows for anonymous reporting of concerns. In addition, the policy describes our commitment to no retaliation associated with reporting of concerns, and the specific follow-on actions to be taken, depending on the nature of any concerns reported through the hotline.
Our marketing efforts are carefully managed, and subject to our Medical, Legal, and Regulatory (MLR) Review process, to ensure that they are in compliance with applicable regulations and the highest ethical standards.
Access to Medicine
Core to our mission and culture is the belief that patients should have access to our medicines and that our staff should be dedicated to serving a patient in need. As a result, we have established and maintain a range of programs designed to maximize access to our medicines. Governance over these programs is led by the Chief Commercial Officer, and program design decisions are reviewed and approved by the Chief Financial Officer and the Chief Executive Officer.
Key programs designed to improve access to our medicines include:
| • | Co-pay discount cards that limit monthly co-pays to $10 for most patients in the United States with commercial insurance. Approximately $6.0 million of co-pay assistance was provided to patients in the United States in 2023. |
| • | A large-scale Patient Assistance Program in the United States, which provides medication at no cost to uninsured or under-insured patients who cannot afford our medication. Approximately 20 percent of patients starting NERLYNX in 2023 received their medication at no cost. |
| • | Participation in the 340B program which provides medications to Medicaid patients at significant discounts. |
| • | A recently-launched pilot program to provide new patients with an initial month of medication at no cost to see if they tolerate the medicine before committing to longer-term use. |
| • | Supportive care medications provided at no cost to patients using our medications. Approximately $130 thousand of vouchers for supportive care medications were provided to patients in 2023. |
| • | Compassionate use to provide access to medicine for individual patients on humanitarian grounds outside of a specific Company-sponsored clinical study. Compassionate use decisions are managed by our Medical Affairs organization. |
Product Quality
Our commitment to quality is demonstrated through our oversight structure, as well as our extensive set of policies, procedures and systems dedicated to quality and quality assurance. Our Quality Assurance organization is independent of our clinical, manufacturing and business operations to ensure that quality personnel act independently and have autonomy from business-driven influences. The head of our Quality Assurance organization meets directly with our Chief Executive Officer with any concerns and findings. Our executive team is held responsible for ensuring that an effective quality management system is in place to ensure the quality of drug products and clinical development programs.
We maintain a Quality System that encompasses an organizational structure, resources, policies, standard operating procedures and work instructions designed to ensure that safe and effective products are manufactured; that clinical trials are conducted in a manner that protects the rights, safety and well-being of trial subjects; and that our company-wide commitment to quality and the performance of the Quality System is supported by our executives and senior management. The main attributes of the Quality System are documented in our Quality Manual, which is a controlled document that cross-references the key policies and standard operating procedures that govern various aspects of the Quality System.
Some of the key aspects of our Quality System include:
| • | A designated person responsible for the quality of drug product released for commercial and clinical use. This person is independent of manufacturing operations and has the authority to approve or reject starting materials, drug substance, bulk product, intermediate product and finished product. |
| • | A designated person responsible for the quality, ethics and compliance of the research, development and clinical programs. This person is independent of research and clinical operations and provides quality and compliance oversight over the clinical studies and other research and development activities. |
| • | An electronic document management system (EDMS) for maintaining regulated documents throughout their lifecycle. |
| • | A validated electronic Learning Management System (LMS) that records evidence of successful completion of training files for all employees and contractors involved in all regulated activities. |
| • | Engagement with our vendor organizations contracted to: manufacture, package, label or test drug products; manage clinical trials; perform clinical site monitoring activities; or perform any other activity that could impact the quality of our products or clinical trials, to address a wide range of issues, including conformance to recognized standards of current Good Manufacturing Practice (cGMP), Good Laboratory Practice (GLP), Good Clinical Practice (GCP) and Good Pharmacovigilance Practice (GVP). Responsibilities for activities related to drug product quality and any critical clinical research services are specified in a separate, written Quality Agreement between us and the vendor. |
| • | External and internal activities that impact the quality of our product or clinical trials are subject to regular audits to ensure compliance with regulatory requirements and our policies, standard operating procedures and work instructions, as applicable. The audits are conducted according to a pre-defined schedule, depending on the nature of the activity. |
Patient and Clinical Trial Safety
We maintain a Safety Review Committee supported by our Pharmacovigilance organization. We have policies and standard operating procedures in place to ensure timely and comprehensive collection, review and reporting of adverse events and serious adverse events in compliance with regulatory requirements in the regions where our products are being studied or marketed. Our Pharmacovigilance organization maintains current information on the drug safety profile of our products as well as all applicable safety-related regulations. Our Pharmacovigilance organization also prepares updates on safety data from clinical trials and post-marketing, scientific literature and safety signals for presentation to the Safety Review Committee. The Safety Review Committee has responsibility for:
| • | Ensuring the execution of strategies aimed at evaluation, validation, and providing recommendations for action of safety risks for our products throughout the development life cycle and following authorization for commercial use. |
| • | Reviewing new safety information as it becomes available. |
| • | Evaluating all available information to validate or refute potential safety signals. Provide recommendations for action, and communication to internal and external stakeholders. |
In addition, standard operating procedures govern the processes that facilitate intake of safety-related inquiries, questions, and reports at any time. Medical review of each individual case safety report is performed by an internal team of medical professionals. Safety information is collected, processed, reported, and maintained in a centralized safety database.
Pharmaceuticals in the Environment
Two of the key pathways by which pharmaceuticals may end up in the environment are through effluent from manufacturing and improper disposal of medications. We contractually require contractors performing manufacturing on our behalf to comply with all federal, state and local laws and regulations regarding effluent to minimize any potential impact through this pathway. With regards to disposal of medications, we participate in MED-Project, a nationwide initiative to promote the proper storage and disposal of medications. MED-Project is run by the Pharmaceutical Product Stewardship Working Group and is designed to facilitate use of local medication disposal and takeback programs. MED-Project operates in all 50 states and maintains a database of over 22,000 locations for disposal and takeback. Through participation in this program, we are promoting proper disposal of unused medication and reducing potential impacts from improper disposal.
Our Board has approved a compensation program for our non-employee directors (the “Director Compensation Program”), which governed their compensation for 2023. The Director Compensation Program is intended to fairly compensate our directors for the time and effort necessary to serve on the Board.
Every other year the Compensation Committee engages Compensia to perform an assessment of the Director Compensation Program relative to its then peer group of companies. In early 2023, Compensia provided their assessment to Company management and the Board and no changes were made to our Director Compensation Program following this review. We expect Compensia to provide an assessment of the program in early 2025, at which time we will evaluate whether any changes should be made to our Director Compensation Program going forward.
Director Compensation Program
Under our non-employee director compensation program effective in 2015, each non-employee director will receive an option to purchase 30,000 shares of our common stock under the Plan upon election or appointment to our Board. In addition, each non-employee director who is appointed to serve on a committee of our Board in a non-chair capacity will receive an option to purchase 10,000 shares of our common stock under the Plan upon appointment and each non-employee director who is appointed to serve as the chair of a committee of our Board will receive an option to purchase 20,000 shares of our common stock upon appointment.
In addition, under the program,Director Compensation Program, each non-employee director receives an annual feecash retainer for service on the Board, and for service on each committee of $50,000, to bewhich the non-employee director is a member, paid in four equal installments of $12,500 at the beginning of each quarter, and each non-employee director who is serving on our Board as of the date of the last regularly scheduled Board meeting held during each calendar year receives an annual option to purchase 10,000 shares of our common stock under the Plan.quarter. All cash fees are payable on a pro-rated basis for non-employee directors who are initially elected or appointed in the middle of a calendar quarter. Each option granted pursuant
The fees paid to non-employee directors for service on the Board under the Director Compensation Program are as follows:
Cash Compensation | ||||
Board Annual Retainer | $ | 50,000 | ||
Committee Chair Annual Retainer | ||||
Audit | $ | 20,000 | ||
Compensation | $ | 15,000 | ||
Nominating and Corporate Governance | $ | 10,000 | ||
Research and Development | $ | 15,000 | ||
Committee Member (Non-Chair) Annual Retainer | ||||
Audit | $ | 10,000 | ||
Compensation | $ | 7,500 | ||
Nominating and Corporate Governance | $ | 5,000 | ||
Research and Development | $ | 7,500 |
In addition, under the Director Compensation Program, each of our non-employee director compensation program will vest overdirectors receives an annual RSU award with a three-year period fromvalue of $300,000 (determined using the trailing 30-calendar day average stock price through and including the grant date) (the “Annual RSU Award”), up to a maximum of 27,000 shares. The 27,000 share limit has resulted in less than $300,000 of equity award value being granted for the past 2 years. The Annual RSU Award is granted automatically on the date of grant, with one-thirdthe annual stockholders’ meeting and vests in full on the earlier of the shares underlying the option vesting on the one-year anniversary of the grant date and then in substantially equal monthly installments over the next two years,date of the annual meeting following the date of grant, subject to the non-employee director’s continued service through the applicable vesting date.
Upon initial appointment or election to the Board, under the Director Compensation Program, each new non-employee director receives a stock option to purchase a number of shares of common stock under the 2011 Plan with a value of $700,000 (determined using a Black-Scholes option value based on a trailing 30-calendar day average stock price), up to a maximum of 100,000 shares. The stock options which vest and become exercisable with respect to one-third of the shares on the first anniversary of the grant date, and with respect to an additional 1/36th of the shares on each monthly anniversary thereafter, subject to the non-employee director’s continued service through the applicable vesting date (the “Initial Option Award”). Each Initial Option Award has an exercise price per share of common stock equal to the fair market value on the date of grant.
Director Compensation
Pursuant to the terms of our 2011 Plan, any compensation payable to our non-employee directors is limited such that the maximum aggregate value of cash compensation and equity-based awards granted to any non-employee director during 2015any calendar year is $1,000,000.
The following table sets forth information regarding the compensation earned by our non-employee directors for the year ended December 31, 2015.2023. Mr. Auerbach, who served as our President and Chief Executive Officer during the year ended December 31, 2015,2023, and continues to serve in that capacity, does not receive additional compensation for his service as a director, and therefore is not included in the Director Compensation table below. All compensation paid to Mr. Auerbach is reported in the Summary Compensation Table included under “Executive Compensation.”
Name | Fees Earned or Paid in Cash ($) | Option Awards ($)(1) | All Other Compensation ($) | Total ($) | ||||||||||||
Thomas R. Malley(2) | 37,500 | — | — | 37,500 | ||||||||||||
Jay M. Moyes(3) | 50,000 | 1,446,478 | — | 1,496,478 | ||||||||||||
Adrian M. Senderowicz(4) | 12,500 | 2,675,453 | — | 2,687,953 | ||||||||||||
Troy E. Wilson(5) | 50,000 | 927,861 | — | 977,861 | ||||||||||||
Frank E. Zavrl (6) | 12,500 | 2,644,542 | — | 2,657,042 | ||||||||||||
Director Compensation Table
Name | Fees Earned or Paid in Cash ($) | Stock Awards ($) (1) | Total ($) | |||||||||
Alessandra Cesano M.D., Ph.D. | 57,500 | 94,770 | 152,270 | |||||||||
Allison Dorval | 60,000 | 94,770 | 154,770 | |||||||||
Michael P. Miller | 68,125 | 94,770 | 162,895 | |||||||||
Jay M. Moyes | 77,500 | 94,770 | 172,270 | |||||||||
Adrian M. Senderowicz, M.D. | 70,000 | 94,770 | 164,770 | |||||||||
Brian Stuglik, R.Ph. | 66,875 | 94,770 | 161,645 | |||||||||
Troy E. Wilson, Ph.D., J.D. | 77,500 | 94,770 | 172,270 | |||||||||
(1) | Represents the grant date fair |
Director Stock Ownership Guidelines
Our corporate governance guidelines contain certain stock ownership guidelines for our non-employee directors. Pursuant to these guidelines, each non-employee director is expected to attain beneficial ownership of 10,000 shares of our common stock within three years after joining the Board, and to maintain or exceed such ownership throughout service as our director. Unless otherwise approved by the Board, each such director must refrain from selling any shares of our common stock (other than for purposes of paying taxes associated with the acquisition of such shares) until the minimum ownership requirement is met. As of December 31, 2023, each non-employee director who had served three years or more on the Board had met these ownership requirements.
SECURITY OWNERSHIP OF DIRECTORS, AND EXECUTIVE OFFICERS AND
CERTAIN BENEFICIAL OWNERS
The following table sets forth the number of shares of our common stock beneficially owned as of April 20, 2016,22, 2024, by (i) each person known by us to be the beneficial owner of more than 5% of the outstanding shares of our common stock, (ii) each of our directors and director nominees, (iii) each of our Named“Named Executive OfficersOfficers” identified under the “Executive Compensation” section of this proxy statement, and (iii)(iv) all current executive officers and directors as a group. Unless otherwise noted below, the address of each stockholder below is c/o Puma Biotechnology, Inc., 10880 Wilshire Boulevard, Suite 2150, Los Angeles, CA 90024.
| SHARES BENEFICIALLY OWNED(1)(2) | ||||||||||
NAME | TITLE | NUMBER(#) | PERCENTAGE | |||||||
Directors and Named Executive Officers | ||||||||||
Alan H. Auerbach(3) | President, Chief Executive Officer and Chairman of the Board | 6,722,915 | 19.1 | % | ||||||
Charles R. Eyler(4) | Senior Vice President, Finance and Administration and Treasurer | 157,999 | * | |||||||
Richard P. Bryce, MBChB, MRCGP, MFPM(5) | Senior Vice President, Clinical Research and Development | 168,382 | * | |||||||
Steven Lo | Chief Commercial Officer | — | — | |||||||
Jay M. Moyes(6) | Director | 83,332 | * | |||||||
Adrian M. Senderowicz, M.D. | Director | — | — | |||||||
Troy E. Wilson(7) | Director | 49,786 | * | |||||||
Frank E. Zavrl(8) | Director | 913,076 | 2.8 | % | ||||||
All executive officers and directors (including nominees) as a group (9 individuals) | 8,095,490 | 22.7 | % | |||||||
Stockholders Holding 5% or More | ||||||||||
Adage Capital Partners L.P.(9) | — | 5,686,668 | 17.5 | % | ||||||
FMR LLC(10) | — | 4,865,362 | 15.0 | % | ||||||
Grantham, Mayo, Van Otterloo & Co. LLC(11) | — | 2,899,776 | 8.9 | % | ||||||
Entities affiliated with OrbiMed Advisors LLC(12) | — | 2,238,825 | 6.9 | % | ||||||
The Vanguard Group, Inc. (13) | — | 1,789,112 | 5.5 | % | ||||||
T. Rowe Price Associates, Inc.(14) | — | 1,719,612 | 5.3 | % | ||||||
Capital Research Global Investors(15) | — | 1,826,600 | 5.6 | % | ||||||
Entities affiliated with Point72 Asset Management, L.P.(16) | — | 1,664,977 | 5.1 | % | ||||||
SHARES BENEFICIALLY OWNED (1) | ||||||||||
NAME | TITLE | NUMBER (#) | PERCENTAGE | |||||||
Directors and Named Executive Officers | ||||||||||
Alan H. Auerbach (2) | President, Chief Executive Officer and Chairman of the Board | 10,259,467 | 20.1 | % | ||||||
Maximo F. Nougues (3) | Chief Financial Officer | 412,664 | * | |||||||
Alvin Wong, Pharm.D. (4) | Chief Scientific Officer | 277,179 | * | |||||||
Jeff J. Ludwig (5) | Chief Commercial Officer | 539,620 | 1.0 | % | ||||||
Douglas Hunt, B.Sc (Hons) (6) | Chief Regulatory Affairs, Medical Affairs, Pharmacovigilance, and Law Officer | 333,071 | * | |||||||
Alessandra Cesano, M.D., Ph.D. (7) | Director | 88,111 | * | |||||||
Allison Dorval (8) | Director | 149,700 | * | |||||||
Michael P. Miller (9) | Director | 60,984 | * | |||||||
Jay M. Moyes (10) | Director | 100,926 | * | |||||||
Adrian M. Senderowicz, M.D. (11) | Director | 104,604 | * | |||||||
Brian Stuglik, R.Ph. (12) | Director | 152,911 | * | |||||||
Troy E. Wilson, Ph.D., J.D. (13) | Director | 75,504 | * | |||||||
All executive officers and directors as a group (12 individuals)(14) | 12,554,741 | 23.7 | % | |||||||
Stockholders Holding 5% or More | ||||||||||
The Vanguard Group, Inc. (15) | 3,802,081 | 7.9 | % | |||||||
Millennium Management, LLC (16) | 3,164,249 | 6.6 | % | |||||||
Acorn Capital Advisors (17) | 2,934,496 | 6.1 | % | |||||||
Camber Capital Management, L.P. (18) | 2,805,000 | 5.8 | % | |||||||
Frazier Life Sciences funds (19) | 2,631,713 | 5.5 | % | |||||||
* | Denotes less than 1.0% of beneficial ownership. |
(1) | Applicable percentages are based on 48,238,850 shares of our common stock outstanding as of April 22, 2024. This table is based upon information supplied by our officers, directors, principal stockholders and transfer agent, and information contained in Schedules 13D and 13G filed with the SEC. Unless otherwise noted in the footnotes to this table, we believe each of the stockholders named in this table has sole voting and investment power with respect to the shares indicated as beneficially owned, subject to community property |
| laws, where applicable. |
| Beneficial ownership is determined in accordance with SEC rules, and includes any shares as to which the stockholder has sole or shared voting power or investment power, and also any shares which the stockholder has the right to acquire within 60 days of April |
(2) | Consists of (i) |
(3) | Consists |
(4) | Consists of (i) 70,327 shares held by Dr. Wong, (ii) options to purchase |
(5) | Consists of (i) 66,484 shares |
(6) | Consists of |
| Consists of | |
(8) | Consists of (i) 17,000 shares held by |
(9) | Consists of (i) 16,358 shares held by |
(10) | Consists of (i) 16,322 shares held by |
(11) | Consists of (i) stock awards of 27,000 shares vesting within 60 days of April 22, 2024 by Dr. Senderowicz, and |
(12) | Consists of (i) 31,958 shares held by Mr. Stuglik, (ii) stock awards of 27,000 shares vesting within 60 days of April 22, 2024, and (iii) options to purchase 93,953 shares of our common stock exercisable within 60 days of April 22, 2024. |
(13) | Consists of (i) stock awards of 27,000 shares vesting within 60 days of April 22, 2024 by Dr. Wilson, (ii) 350 shares held in an IRA by Dr. Wilson, (iii) a total of 550 shares held in minor accounts for Dr. Wilson’s children, and (iv) options to purchase 47,604 shares of our common stock exercisable within 60 days of April 22, 2024. |
(14) | With respect to the |
(15) | The information reported is based on a Schedule 13G/A filed on February 13, 2024, reporting ownership as of December 29, 2023. The Vanguard Group, Inc. reports sole dispositive power with respect to 3,787,638 shares of our common stock and shared dispositive power with respect to 14,443 shares of our common stock. The address for this entity is 100 Vanguard Blvd., Malvern, PA 19355. |
(16) | The information reported is based on a Schedule 13G/A filed on January 30, 2024 reporting ownership as of December 31, 2023. Integrated Core Strategies (US) LLC reports shared voting power and shared dispositive power with respect to 2,843,239 shares. Millennium Management LLC, Millennium Group Management LLC and Israel A. Englander each report shared voting power and shared dispositive power with respect to 3,164,249 shares of our common stock, and that such shares are held by entities subject to voting control and investment discretion by Millennium Management LLC and/or other investment managers that may be controlled by Millennium Group Management, LLC (the managing member of Millennium Management LLC) and Mr. Englander (the sole voting trustee of the managing member of Millennium Group Management LLC). The address for these entities and person is 399 Park Avenue, New York, NY 10022. |
(17) | The information reported is based on a Schedule 13G/A filed on February |
(18) | The information reported is based on a Schedule 13G/A filed on February |
| The information reported is based on a Schedule 13G filed on |
Set forth below is information regarding each of our executive officers as of April 22, 2024, the record date.
Name | Age | Position |
Alan H. Auerbach | 54 | President, Chief Executive Officer and | ||
Maximo F. Nougues | 55 | Chief Financial Officer | ||
Alvin Wong, Pharm.D. | 71 | Chief Scientific Officer | ||
Jeff J. Ludwig | 58 | Chief Commercial Officer | ||
Douglas Hunt, B.Sc (Hons) | 59 | Chief Regulatory Affairs, Medical Affairs, Pharmacovigilance, and |
Alan H. Auerbach. See “Director Biographical Information” above.
Maximo F. Nougues. Mr. Nougues joined the Company as Chief Financial Officer in November 2018. Prior to joining the Company, Mr. Nougues served in various positions at Getinge AB, a global medical device company based in Sweden, from January 2008 until October 2018. At Getinge, he held several leadership positions with oversight for the business that generated regional revenues of approximately $1 billion annually. During his tenure at Getinge, he served as Regional Chief Financial Officer for North America, Regional Chief Financial Officer for the Americas and Regional Vice President of Finance. Mr. Nougues also served as Chief Financial Officer for MAQUET North America, which was acquired by Getinge in 2000. Prior to joining Getinge, Mr. Nougues worked in finance roles in Boston Scientific’s cardiac surgery division, which was acquired by Getinge in 2008, and at The Clorox Company from 1998 until 2007. Mr. Nougues holds an M.S. in business administration from the Universidad del Norte Santo Tomas de Aquino, Tucuman, Argentina and an M.B.A. from the University of San Francisco McLaren School of Business.
Alvin Wong, Pharm.D. Dr. Wong joined the Company in March 2013 and since June 2021 has served as our Chief Scientific Officer. From August 2017 to June 2021, Dr. Wong served as our Senior Vice President, Clinical Science and Pharmacology. From 2007 to 2013, Dr. Wong was Senior Director of Clinical Development at Proteolix, Inc. (acquired by Onyx) where he managed studies in multiple myeloma and supported the approval of Kyprolis®. Previously, he held positions in Clinical Pharmacology and Pharmacovigilance at Novacea, and in drug safety at Genentech, where he contributed to the successful submissions of Herceptin® for HER2-positive breast cancer, Rituxan® for patients with non-Hodgkin's Lymphoma and Avastin® for patients with colorectal cancer. Dr. Wong earned his Doctor of Pharmacy degree from the University of San Francisco (UCSF). Dr. Wong was a Clinical Professor at UCSF from 1976 to 1996.
Jeff J. Ludwig. Mr. Ludwig has served as our Chief Commercial Officer since March 2020. Mr. Ludwig previously worked as Vice President Oncology Sales for Astellas Pharma Inc. (“Astellas”), a multinational pharmaceutical company based in Japan, from September 2019 until March 2020, where he led multiple sales teams across several oncology therapeutic products, a significant label expansion in prostate cancer and a new launch in bladder cancer. Prior to joining Astellas, Mr. Ludwig worked at Amgen, a multinational biopharmaceutical company, from April 2001 until August 2019, where he most recently held the position of Vice President of Oncology Sales from January 2014 to August 2019. During his tenure as Vice President of Oncology Sales, he led the sales organization through numerous successful launches and contributed to a multi-billion dollar portfolio of therapeutic and supportive care products. Before assuming the Vice President of Oncology Sales role, he successfully held various other leadership positions at Amgen in both oncology sales and marketing. Prior to joining Amgen, Mr. Ludwig worked in various sales and marketing roles at Eli Lilly and Company from 1988 until 2001. Mr. Ludwig holds a B.S. in business from Arizona State University where he graduated with honors.
Douglas Hunt B.Sc (Hons) Mr. Hunt has served as our Chief Regulatory Affairs, Medical Affairs, Pharmacovigilance, and Law Officer since April 2024. Mr. Hunt commence employment in January 2018 as Senior Vice President, Regulatory Affairs and has been responsible for various operations of the Company during his tenure, including Pharmacovigilance since 2023, Medical Affairs since 2022, and Law since 2020. Previously, Mr. Hunt also oversaw the Company’s Medical Writing and Project Management. Mr. Hunt has over 25 years of regulatory affairs experience and was a regulatory affairs consultant to the Company from February 2017 to January 2018. Mr. Hunt previously served as Vice President Regulatory Affairs and Quality Assurance at ArmaGen, Inc., a private biotechnology company, from March 2015 until December 2017 and Vice President, Global Regulatory Affairs (Bioscience) at Baxter International Inc., a public healthcare company, from March 2008 March 2015 where he was responsible for global regulatory affairs for several franchises including oncology. Prior to his role at Baxter Bioscience, Mr. Hunt worked for Amgen, a multinational biopharmaceutical company, from June 2000 to March 2008 in various positions, including as Executive Director, Therapeutic Area Head (Oncology) and Executive Director, Therapeutic Area Head (Bone/Oncology), Global Regulatory Affairs and Safety. Mr. Hunt received a B.Sc (Hons). from Portsmouth University.
Compensation Discussion and Analysis
Executive Summary
The Compensation Committee of our Board develops our executive compensation policies and determines the amounts and elements of compensation for our named executive officers (“Named Executive Officers”).
We have in place a compensation strategy for our executives that we believe focuses on both individual and Company performance. Incentive compensation paid to our executives is awarded based on our Compensation Committee’s review of our achievement of near-term corporate targets and longer term business objectives and strategies. The Compensation Committee is responsible for evaluating and administrating all of our executive compensation programs and practices to ensure that they properly compensate, reward and drive corporate performance while remaining competitive with comparable biotechnology companies. The Compensation Committee reviews and approves all compensation for our executive officers, including base salaries, annual bonuses and equity incentive compensation.
This Compensation Discussion and Analysis describes our executive compensation programs for our Named“Named Executive OfficersOfficers” for the 20152023 fiscal year, who were:
| • | Alan H. Auerbach, our President and Chief Executive Officer; |
| • | Maximo F. Nougues, our Chief Financial Officer; |
| • | Alvin Wong, Pharm.D., our Chief Scientific Officer; |
| • | Jeff J. Ludwig, our Chief Commercial Officer; and |
| • | Douglas Hunt, B.Sc (Hons), our Chief Regulatory Affairs, Medical Affairs, Pharmacovigilance, and Law Officer. |
Alan H. Auerbach, our President and Chief Executive Officer;
Charles R. Eyler, our Senior Vice President, Finance2023 Business Highlights
| • | 2023 Financial Performance. Our revenue, net income and cash flow improved in 2023. Total revenue for the year ended December 31, 2023 improved to approximately $235.6 million, compared to approximately $228.0 million for the year ended December 31, 2022. Net income for the year ended December 31, 2023 improved to approximately $21.6 million, or basic net income per share of $0.46 and diluted net income per share of $0.45, compared to net income of two thousand dollars, or $0.00 per share basic and diluted, for the year ended December 31, 2022. Additionally, net cash flow increased by $7.9 million for the year ended December 31, 2023, compared to an increase of $3.5 million for the year ended December 31, 2022. |
The below table summarizes the financial results of the Company over the years ended December 31, 2023, 2022 and Administration2021 (in thousands except share and Treasurer;per share data):
Richard P. Bryce, MBChB, MRCGP and MFPM, our Senior Vice President, Clinical Research and Development; and
For the Year Ended December 31, | ||||||||||
| 2023 | 2022 | 2021 | ||||||||
| Product revenue, net | $ | 203,107 | $ | 200,023 | $ | 189,064 | ||||
| License revenue | — | — | 51,750 | |||||||
| Royalty revenue | 32,530 | 28,008 | 12,341 | |||||||
Total revenue | $ | 235,637 | $ | 228,031 | $ | 253,155 | ||||
Income from operations | $ | 32,640 | $ | 23,720 |
| $ | 1,290 | |||
Net income (loss) | $ | 21,591 |
| $ | 2 |
| $ | (29,126 | ) | |
Net income (loss) applicable to common stockholders | $ | 21,591 |
| $ | 2 |
| $ | (29,126 | ) | |
Net income (loss) per share of common stock—basic | $ | 0.46 | $ | 0.00 |
| $ | (0.72 | ) | ||
| Net income (loss) per share of common stock—diluted | $ | 0.45 | $ | 0.00 | $ | (0.72 | ) | |||
| • | Alisertib Exclusive License. 2023 marked the first year in which alisertib was part of operations, having acquired an exclusive worldwide license for the research and development of, and commercial rights to, alisertib in September 2022. Alisertib is an investigational, reversible, ATP-competitive inhibitor that is designed to be highly selective for aurora kinase A. Inhibition of aurora kinase A can lead to disruption of mitotic spindle apparatus assembly, disruption of chromosome segregation, and inhibition of cell proliferation. In clinical trials to date, alisertib has shown single agent activity and activity in combination with other cancer drugs in the treatment of many different types of cancers, including hormone receptor-positive breast cancer, triple negative breast cancer, small cell lung cancer and head and neck cancer. |
Steven Lo, our Chief Commercial Officer.
2023Compensation Highlights
| • | Achievement of 2023 Corporate Performance Goals – Below Target Payout Despite Improved Revenue and Net Income. Our compensation philosophy is designed to align the interests of our executive officers with those of our stockholders by providing pay that is directly linked to the achievement of performance goals established to foster the creation of sustainable long-term stockholder value and year-over-year improvement. As such, the portion of our 2023 short-term incentive compensation program for our Named Executive Officers, which was based on previously established goals related to total revenue (excluding license revenue), cashflow, alisertib clinical trial and alisertib product development, were deemed achieved at 49.0%, 73.2%, 50% and 150% of target for that goal, respectively, which resulted in an overall 71.5% attainment of the corporate performance portion of the annual short-term incentive compensation award opportunity, after weighting, as described further below. The revenue goal was weighted 40%, the net income goal was weighted 30%, and each of the two alisertib goals were weighted 15% each. The revenue and net income threshold goals were set at last year’s actual performance, with at-threshold performance resulting in 0% earned for that goal and linear interpolation to performance at target, which yields 100% payout, and maximum performance, which yields 200% payout, for that goal. Mr. Auerbach’s short-term incentive compensation is based 100% on our corporate performance and thus he was awarded 2023 short-term incentive compensation of 71.5% of target opportunity. For the other Named Executive Officers (other than Mr. Auerbach), after taking into account the Compensation Committee’s assessment of each executive’s individual performance and the performance of the operations for which the executive is responsible, the 2023 short-term incentive compensation awarded to Messrs. Nougues, Hunt and Ludwig and Dr. Wong was 88.25%, 88.25%, 68.25% and 83.25%, respectively, of each executive’s target bonus opportunity, based on individual performance assessed at 105%, 105%, 65%, and 95% of target, respectively. |
| • | Increased 2023 Equity Award Values Based on Company Performance and Market Median. The long-term equity values granted for 2023 reflect the Compensation Committee’s general approach in determining annual equity grant value, wherein the median long-term equity value granted at our peer group for the similar position is modified by the Company’s actual performance under the corporate goals for our just completed prior year’s annual short-term incentive compensation program. Thus, in 2023, the primary factors that impacted the value of long-term equity-based incentive awards granted was our 158.4% performance against corporate goals for 2022 under our annual short-term incentive compensation program and the 50th percentile of the long-term equity values of our 2023 peer group. In approving these increases, the Compensation Committee also reviewed and considered 2023 peer group benchmarking data that reflected the prior year’s equity grant values for our NEOs being significantly below market median, the recommendations of our CEO for an adjustment in grant value for the other NEOs, individual performance by each of the NEOs, and the limitation imposed by the shares available for grant under our 2011 Plan. These additional considerations resulted in 2023 equity grant values for each executive at 149% of peer group median for their position. Under this formula, the total value of the 2023 long-term equity-based incentive award for Mr. Auerbach increased from approximately $790,000 in 2022 to approximately $1.6 million in 2023, which methodology aligns with our pay for performance philosophy. |
Advisory Vote on Executive Compensation
At our 20152023 annual meeting of stockholders, we held a say-on-pay vote, andapproximately 93% of the votes cast by our stockholders approved, on an advisory basis, the compensation of our Named Executive Officers, as disclosed in the proxy statement for that meeting pursuant to the compensation disclosure rules of the SEC. Upon review of the final voting results, and given the significant level of stockholder support (approximately 91% of the votes cast approved the say-on-pay proposal), we haveOfficers. We did not mademake any changes to our executive compensation policies or decisions as a result of the vote. In addition,As described in further detail under Proposal 4 of this proxy statement, at our 2012this annual meeting, of stockholders, we held a say-on-frequency vote and our stockholders indicated their preference for three years as the frequency of futureare being asked to indicate how frequently they believe we should seek an advisory votesvote on the compensation of our Named Executive Officers. Accordingly,Officers (the “frequency vote”). Our Board has recommended that our stockholders vote for a frequency of one year. We currently expect our next say-on-pay vote (after the vote on Proposal 3 at this annual meeting) will be held at our 2025 annual meeting, although our Board may decide to modify this expectation, particularly in 2018.light of the results of the frequency vote.
2023 Shareholder Engagement
We value the opinions of our stockholders on our executive compensation programs. In evaluating the design of our executive compensation, the Compensation Committee considers stockholder input, which we solicit annually through direct outreach with our large investors and through our annual advisory say-on-pay vote. In 2023, we gathered feedback from our stockholders on topics including our compensation program, board composition, board expertise and diversity and over-boarding.
Chief Executive Officer Realized Compensation
The graph below illustrates the Summary Compensation Table reported values for Mr. Auerbach’s total compensation for 2022 and 2023 in the Summary Compensation Table and his total direct compensation realized for each year. We believe this graph shows that Mr. Auerbach’s total direct compensation realized is less than the Summary Compensation Table-reported values for 2022 and 2023, and that the compensation reported in the Summary Compensation Table overstates Mr. Auerbach’s compensation.
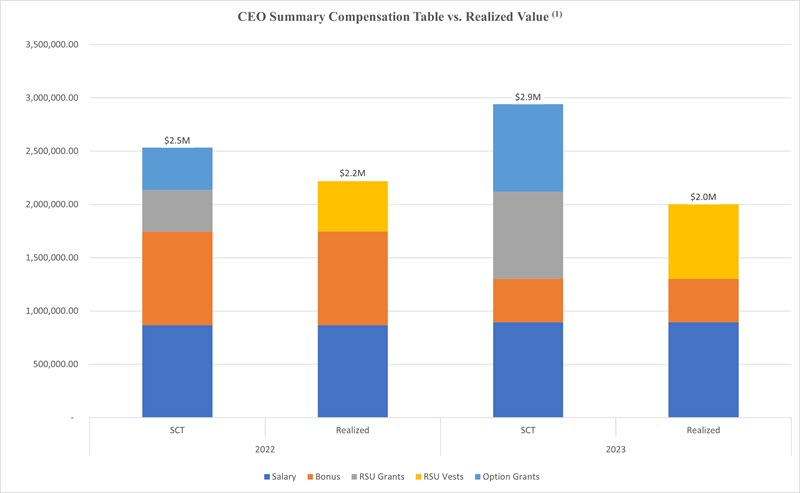
(1) For purposes of this graph, realized value includes Mr. Auerbach’s salary and short-term incentive compensation for the applicable year, as well as benefits and other perquisites, the value of any RSUs that vested, and any stock options exercised in the applicable year.
At December 31, 2022 and 2023, all of the outstanding options held by our executive officers were underwater with exercise prices in excess of the closing stock price of our common stock on December 30, 2022 and December 29, 2023 (the last trading days of the applicable calendar year) of $4.23 and $4.33 per share, respectively, except for the option grants to our executives on March 3, 2022 at an exercise price of $2.33 per share.
Compensation Principles and Objectives
Our overall compensation program is structured to attract, motivate and retain highly qualified executive officers by paying them competitively, consistent with our success and their contribution to that success. Our ability to excel depends on the skill, creativity, integrity and teamwork of our employees. Given the long product development cycles in our business, we believe compensation should be structured to ensure that a portion of compensation opportunity will be related to factors that directly and indirectly influence long-term stockholder value. Our compensation philosophy has been driven by a number of factors that are closely linked with our broader strategic objectives.
The Compensation Committee believes that compensation paid to our Named Executive Officers should be aligned with our performance on both a short-term and long-term basis, linked to results intended to create value for stockholders, and that such compensation should assist us in attracting and retaining key executives critical to our long-term success.
In establishing compensation for executive officers, the following are the Compensation Committee’s objectives:
align officer and stockholder interests by providing a portion of total compensation opportunities for senior management in the form of equity awards and bonuses awarded based on the Compensation Committee’s review of company and individual performance;
| • | Align officer and stockholder interests by providing a portion of total compensation opportunities for senior management in the form of equity awards and short-term incentive compensation awarded based on the Compensation Committee’s review of Company and individual performance; |
| • | ensure executive officer compensation is competitive within the marketplace in which we compete for executive talent by relying on the Compensation Committee’s judgment, expertise and personal experience with other similar companies, recognizing that because of the Company’s business model and relatively early stage of development, there may be few directly comparable companies; and |
| • | recognize that best compensation practices for a growing company may be substantially different than for a larger, more mature company and that we should make full use of our greater latitude and breadth of compensation opportunities. |
Strong Governance and Compensation Practices
We endeavor to maintain sound governance standards consistent with our executive compensation policies and practices. The Compensation Committee evaluates our executive compensation program on a regular basis to ensure that it is consistent with our short-term and long-term goals given the dynamic nature of our business and the market in which we compete for executive talenttalent. Our compensation philosophy and related corporate governance policies and practices are complemented by relying on the Compensation Committee’s judgment, expertise and personal experience with other similar companies, recognizing that because of the Company’s business model and relatively early stage of development, there may be few directly comparable companies; and
recognize that bestfollowing specific compensation practices for a growing company may be substantially different than for a larger, more mature company and that we should make full use ofare designed to align our greater latitude and breadth ofexecutive compensation opportunities.program with long-term stockholder interests:
What We Do | What We Do Not Do | ||
✓ | Multi-Year Vesting. The equity awards granted to our executive officers generally vest over multi-year periods, which we believe is consistent with current market practice, our retention objectives and our pay for performance philosophy. | ✕ | No Guaranteed Compensation. We do not guarantee cash incentives, equity compensation or salary increases for executive officers. |
✓ | Stock Ownership Guidelines. We maintain executive stock ownership guidelines that cover our executive officers. | ✕ | No “Single-Trigger” Compensation. We have no single-trigger cash severance, equity acceleration or other benefits in connection with a change in control; instead, payments require a termination of employment in connection with the change in control. |
✓ | Clawback Policy. In 2023, we updated our clawback policy to recoup erroneously-paid cash and equity compensation from our executive officers in the event of a financial restatement. | ✕ | Hedging and Pledging Prohibited. We prohibit our executive officers and the non-employee members of our Board from hedging or pledging our securities. |
✓ | Long-Term Incentive Awards. The majority of our executive officers' compensation is granted in the form of long-term equity awards, which we believe aligns their interests with those of our stockholders. | ✕ | No Repricing without Stockholder Approval. Our equity plan prohibits repricing of out-of-the-money stock options to a lower exercise price without approval of our stockholders. |
✓ | Limited Perquisites. We provide only limited perquisites or other personal benefits to our executive officers, such as Company-paid long-term disability insurance and annual physicals. | ||
✓ | Independent Compensation Committee. The Compensation Committee is comprised solely of independent directors. | ||
✓ | Independent Compensation Advisor. The Compensation Committee engages an independent compensation consultant to provide independent analysis, advice and guidance on executive compensation. |
Determination of Compensation
The Compensation Committee is charged with the primary authority to determine and recommend the compensation awards available toof our executive officers for approval by the Board.Named Executive Officers. Based on the Compensation Committee members’ collective understanding of compensation practices in similar companies in the biotechnology and pharmaceutical industry, and based upon the advice and recommendation from Compensia (its independent compensation consultant), our executive compensation package consists of the following primary elements, in addition to the employee benefit plans in which all employees may participate:
Base salary: compensation for ongoing services throughout the year.
| • | Base salary: compensation for ongoing services throughout the year. |
Annual discretionary cash bonus awards: discretionary awards to recognize and reward achievement of corporate and individual performance.
| • | Annual short-term incentive compensation awards: awards to recognize and reward achievement of short-term corporate and individual performance. |
Long-term equity incentive program: equity compensation to provide an incentive to our Named Executive Officers to manage us from the perspective of an owner with an equity stake in the business.
| • | Long-term equity incentive awards: equity compensation to provide an incentive to our Named Executive Officers to focus on long-term performance and to manage us from the perspective of an owner with an equity stake in the business. |
| • | Severance and change in control benefits: remuneration available to certain Named Executive Officers in the event of a qualifying termination of employment. |
Severance and change in control benefits: remuneration paid to certain executives in the event of a qualifying termination of employment.
To aid the Compensation Committee in making its determination, our Chief Executive Officer provides recommendations annually to the Compensation Committee regarding the compensation of all other executive officers (other than himself) based on the overall corporate achievements during the period being assessed and his knowledge of the individual contributions to our success by each of the Named Executive Officers. The overall performance of our Named Executive Officers as a team is reviewed annually by the Compensation Committee.
We set base salary and annual bonusshort-term incentive compensation structures and any grants of stock options or RSUs based on the Compensation Committee members’ collective understanding of compensation practices in the biotechnology and pharmaceutical industry and such members’ experiences as seasoned executives, consultants, board and Compensation Committeecompensation committee members, or investors in similar biotechnology and specialty pharmaceutical industry companies.
In addition,November 2022, the Compensation Committee reviewed and revised the composition of our peer group, employing an assessment of our peer group prepared by, and after consideration of the advice of, Compensia, for use as a reference when setting our 2023 executive compensation levels, resulting in establishing the following 16 peer group companies (the “2023 peer group”):
| AcelRx Pharmaceuticals, Inc.(1) | Eagle Pharmaceuticals | Paratek Pharmaceuticals, Inc.(1) |
| AIM ImmunoTech Inc.(1) | Eiger BioPharmaceuticals, Inc.(1) | Rigel Pharmaceuticals |
| Akebia Therapeutics | Fortress Biotech, Inc.(1) | scPharmaceuticals Inc.(1) |
| Alimera Sciences, Inc.(1) | Karyopharm Therapeutics | Trevena, Inc.(1) |
BioXcel Therapeutics, Inc.(1) | MacroGenics, Inc.(1) | |
| Citius Pharmaceuticals, Inc.(1) | OptiNose, Inc.(1) |
(1) | Peer company was added for 2023. |
The market data used for Compensia’s comparative analysis was drawn from timepublicly available sources and also from the Radford Global Life Science Survey. The 2023 peer group was developed with primary consideration given to time we may relycompanies with lower market capitalization than the 2022 peer group, and companies more similar to ours with respect to development phase revenue, market capitalization and employee headcount. Based on the peer analysis performed by Compensia in November 2022 for the purpose of selecting our 2023 peer group, 12 companies were added to the 2023 peer group (as noted above) and the following 15 companies were removed: Adamas Pharmaceuticals; ADMA Biologics; Aerie Pharmaceuticals; BioDelivery Sciences International; Catalyst Pharmaceuticals; Clovis Oncology; Coherus BioSciences; Collegium Pharmaceutical; Flexion Therapeutics; Intercept Pharmaceuticals; La Jolla Pharmaceutical Company; Omeros; Radius Health; Theravance Biopharma; Travere Therapeutics; and Vanda Pharmaceuticals. In general, the removed peer companies no longer fit the selection criteria, while the additions reflect companies with financial and industry characteristics more similar to our company.
In assessing our peer group, Compensia prepared for the Compensation Committee a peer group using the following criteria:
| • | Biotechnology companies with a therapeutic focus and that are in a pipeline stage; |
| • | Peer companies with revenues ranging from $0 to $297 million (with Puma revenues at $207 million or the 83rd percentile), market capitalization ranging from $27 million to $399 million (with Puma market capitalization at $106 million or the 33rd percentile); and |
| • | Peer companies headquartered in the United States and other similar characteristics such as revenue, market cap, one-year and three-year total shareholder return and employee headcount. |
We believe these characteristics are relevant because their similarity captures the similarity in the labor market for executives.
In November 2023, the Compensation Committee reviewed and revised the composition of our peer group, employing an assessment of our peer group prepared by, and after consideration of the advice of, Compensia, for use as a reference when setting our 2024 executive compensation surveylevels, resulting in the following 16 peer group companies (the “2024 peer group”):
Agenus Inc. (1) | Eiger BioPharmaceuticals, Inc. | MacroGenics, Inc. |
Assertio Holdings, Inc. (1) | Esperion Therapeutics (1) | Nektar Therapeutics (1) |
| Akebia Therapeutics | G1 Therapeutics, Inc. (1) | OptiNose, Inc. |
Alimera Sciences, Inc. | Heron Therapeutics (1) | Rigel Pharmaceuticals |
BioXcel Therapeutics, Inc. | Intercept Pharmaceuticals (1) (2) | scPharmaceuticals Inc. |
Eagle Pharmaceuticals | Karyopharm Therapeutics | Theravance Biopharma (1) |
| Y-mAbs Therapeutics (1) |
(1) | Peer company was added in 2024. |
| (2) | Acquired by Alfasigma in November 2023. |
The market data providedused for Compensia’s comparative analysis was drawn from publicly available sources for the companies in the 2023 peer group and also from the Radford Global Life Science Survey. The 2024 peer group was developed with primary consideration given to companies with similar market capitalization as well as removing any pre-commercial companies from the 2024 peer group. Based on the peer analysis performed by Radford or other publicly-available datasets, suchCompensia in 2023 for the purpose of selecting our 2024 peer group, 9 companies were added as from IBM or PayScale.noted above and the following 7 companies were removed: AcelRx Pharmaceuticals, Inc.; AEIM ImmunoTech Inc.; Citius Pharmaceuticals, Inc.; Fortress Biotech, Inc.; MacroGenics, Inc.; Paratek Pharmaceuticals, Inc.; and Trevena, Inc. In general, the removed peer companies no longer fit the selection criteria while the additions reflect companies with financial and industry characteristics more similar to our company.
In assessing our peer group, Compensia prepared for the Compensation Committee a peer group using the following criteria:
| • | Biotechnology companies with a therapeutic focus and that are in a pipeline stage; |
| • | Peer companies with revenues ranging from $75 million to $690 million (with Puma revenues at $230 million or the 97th percentile), market capitalization ranging from $40 million to $590 million (with Puma market capitalization at $146 million or the 57th percentile); and |
| • | Peer companies headquartered in the United States and other similar characteristics such as revenue, market cap, one-year and three year total shareholder return and employee headcount. |
We believe these characteristics are relevant because their similarity captures the similarity in the labor market for executives.
Elements of Executive Compensation
Base Salaries
Mr. Auerbach is
Our Chief Executive Officer periodically reviews and recommends the founder of our company, and accordingly his compensation was initially established to reflect his position as a founding executive and has evolved as we have grown. Mr. Eyler, Dr. Bryce and Mr. Lo joined us after we were founded, and their initial compensation was the result of arms-length negotiations at that time.
Basebase salaries in effect for each of our Named Executive Officers (other than our Chief Executive Officer) are recommendedfor himself), and reviewed periodically by our Chief Executive Officer, andthe Compensation Committee approves the base salary for each Named Executive Officer is approved by our Compensation Committee.Officer. Adjustments to base salaries are based on the scope of an executive’s responsibilities, individual contribution, experience, and sustained performance. Decisions regarding salary increases may take into account the Named Executive Officer’s current salary, equity ownership and the amounts paid to individuals in comparable positions at our peer companies.from the assessment prepared by Compensia. No formulaic base salary increases are provided to our Named Executive Officers. This strategy is consistent with our intent of offering compensation that is cost-effective, competitive and contingent ontakes into account the achievement of performance objectives.executive’s performance.
In 2015,
Effective January 1, 2023, the Compensation Committee approved base salary increases of approximately 3% for Mr. Auerbach, approximately 4% for Messrs. AuerbachLudwig and Eyler, each effective September 1, 2015,Nougues, and Dr. Bryce, effective June 20, 2015. Wong, and approximately 7.5% for Mr. Hunt. These base salary increases reflect performance as well as cost of living increases, which positioned the base salaries in the 80th to 85th percentile of our 2023 peer group.
Effective January 1, 2024, the Compensation Committee approved base salary increases of approximately 3% for Mr. Auerbach, approximately 4.5% for Messrs. Nougues, Hunt, and Dr. Wong, and approximately 2% for Mr. Ludwig.
The following table shows these executives’ annualthe Named Executive Officers’ base salary prior tosalaries for 2022, 2023 and after the increases.2024.
2022 Annual Base | 2023 Annual Base | 2024 Annual Base | ||||||||||||||||||
Name | Prior Annual Base Salary ($) | New Annual Base Salary ($) | Salary ($) | Salary ($) | Salary ($) | |||||||||||||||
Alan H. Auerbach | 630,000 | 693,000 | 852,302 | 877,871 | 904,207 | |||||||||||||||
Charles R. Eyler | 333,108 | 372,600 | ||||||||||||||||||
Richard P. Bryce, MBChB, MRCGP, MFPM | 357,210 | 385,787 | ||||||||||||||||||
Maximo F. Nougues | 495,693 | 515,521 | 538,719 | |||||||||||||||||
Alvin Wong, Pharm.D. | 504,000 | 524,160 | 547,747 | |||||||||||||||||
Jeff J. Ludwig | 573,623 | 596,568 | 608,499 | |||||||||||||||||
Douglas Hunt, B.Sc (Hons) | 398,164 | 428,026 | 447,287 | |||||||||||||||||
The Compensation Committee determined to approve these base salary increases to reward the executives for their significant contributions to the development of the Company, as well as to bring their salaries in line with competitive positions (as determined based on the Compensation Committee’s review of the various compensation survey data mentioned above).
The actual base salaries paid to all of our Named Executive Officers during 20152023 are set forth in the “Summary Compensation Table” below.
Bonuses
Annual Bonuses.Short-term Incentive CompensationCash bonuses are
Our short-term incentive compensation program is intended to provide incentives to drive company-wide performance. Each of our Named Executive Officers is eligible to receive a discretionary cash bonusincentive award targeted as a percentage of the executive’s base salary. For 2015,2023, as in prior years, Messrs. Auerbach, Eyler and Lo,Nougues, Ludwig, Hunt, and Dr. BryceWong were eligible to receive a discretionary cash bonusincentive award targeted at 50%, 35%65%, 40%, 40%, 40%, and 35%40%, respectively, of their annual base salary (andin effect at the end of 2023.
The Company’s 2023 annual short-term incentive compensation program was based in part on the achievement of corporate performance objectives, which represented 100% of the award opportunity for Messrs.Mr. Auerbach, and Eyler,for the other Named Executive Officers, corporate performance objectives represented 50% of the award opportunity and Dr. Bryce,individual performance represented the remaining 50% of the award opportunity. Short-term incentive award opportunities relating to corporate performance objectives (the “Corporate Performance Portion”) were based on the achievement of four performance criteria: (1) total revenue, less license revenue, (2) cashflow, (3) alisertib clinical trial development and (4) alisertib product development.
Award opportunities for each metric were established at threshold, target and maximum levels of performance, and achievement at threshold (and below) pays out at 0%, achievement at target pays out at 100%, and achievement at or above maximum performance pays out at 200%, of each executive’s target award opportunity. With respect to the revenue and cashflow metrics, straight-line interpolation was used to calculate payouts associated with actual results falling between threshold and target goals (0-100%), and target and maximum goals (100-200%). Each of the corporate performance objectives are explained in detail as follows:
FINANCIAL PERFORMANCE METRIC | WEIGHTING | THRESHOLD 0% Payout | TARGET 100% Payout | MAXIMUM 200% Payout | ACTUAL | ATTAINMENT FACTOR -% OF TARGET PAYOUT (AFTER WEIGHTING) | |
Total revenue, less license revenue Sum of (a) total product revenue, net and (b) royalty revenue. The goals were rigorous with threshold goal approximately equal to 2022 actual performance. | 40% | $228.0 million | $243.6 million | $259.1 million | $235.6 million | 49% Attainment 19.6% Payout | |
Cashflow Change from 12/31/22 to 12/31/23 in total cash and cash equivalents, marketable securities, and restricted cash, less (i) significant one-time cash outflows related to lawsuit settlements or asset acquisitions and (ii) capital raises. | 30% | $26.1 million | $38.1 million | $50.1 million | $34.9 million | 73.2% Attainment 22% Payout | |
CLINICAL PERFORMANCE METRIC | WEIGHTING | THRESHOLD 50% Payout | TARGET 100% Payout | PRE-MAXIMUM 150% Payout | MAXIMUM 200% Payout | ACTUAL | ATTAINMENT FACTOR -% OF TARGET PAYOUT (AFTER WEIGHTING) |
Alisertib Clinical Trial Development Total number of alisertib trial site openings and trial patients during 2023. | 15% | Trial Sites: 2 Trial Patients: 0 | Trial Sites: 0 Trial Patients: 1 | Trial Sites: 5 Trial Patients: 3 | Trial Sites: 5 Trial Patients: 5 | Trial Sites: 4 Trial Patients: 0 | 50% Attainment 7.5% Payout |
Alisertib Product Development Drug re-supply and analytical samples testing and release. | 15% | Re-supply incomplete, expiry extension obtained. | Re-supply and testing complete. | Re-supply released by December 31, 2023 | Re-supply released by November 30, 2024 | Completed December 2023 | 150% Attainment 22.5% Payout |
When the Compensation Committee established the goals for 2023 in early 2023, they believed the goals were rigorous given that any payout under the financial metrics required achieving at a minimum actual performance for 2022, and target performance was informed by the Board-established budget for 2023. Under the plan design, performance between threshold and target is interpolated between 0 and 100%. Based on each of the previously defined corporate performance criteria cited above, the final achievement of target opportunity for revenue, cashflow, alisertib clinical trial development and alisertib product development was 49.0%, 73.2%, 50% and 150%, respectively, prior to weightings, which resulted in an overall 71.5% payout of target opportunity after weighting under the 2015 increases).
The determinationCorporate Performance Portion of the amount of annual bonuses paid toshort-term incentive compensation award opportunity.
For our Named Executive Officers, generally reflectsother than Mr. Auerbach, the 2023 short-term incentive compensation program also contained a numberdiscretionary element based on the Compensation Committee’s assessment of considerations bythe executive’s individual performance (the “Individual Performance Portion”), weighted 50%, with the Corporate Performance portion weighted 50%. In assessing our executives’ performance in 2023 at fiscal year-end, the Compensation Committee acting inconsidered their discretion, including, among other things,efforts with respect to the commercial performance of NERLYNX both within and outside of the United States, as well as clinical, financial, and working capital achievements. Among those achievements are: initial development of alisertib, effective management of operating expenses, and a significant improvement of our net income in 2023. Additionally, the Company was cashflow positive for 2023. Based on our Compensation Committee’s consideration of each executive’s individual performance for 2023, with feedback from our Chief Executive Officer, Messrs. Nougues and a subjective evaluationHunt each received 105%, Dr. Wong received 95%, and Mr. Ludwig received 65% of the individual contributiondiscretionary portion of their 2023 short-term incentive compensation opportunity given the contributions each person made in the achievement of the Company’s overall results described above. Each executive’s blended performance for 2023 was as follows: Messrs. Nougues and performanceHunt each received 88.25%, Dr. Wong received 83.25%, and Mr. Ludwig received 68.25% of each executive’s applicable target bonus opportunity.
The following table sets forth the short-term incentive compensation awards paid to each Named Executive Officer. Bonus determinations are not formulaic and no particular weight is assigned to any of the factors considered by the Compensation Committee.Officer for 2023.
In 2015, in accordance with the approach described above, the Compensation Committee awarded cash bonuses of $315,000 to Mr. Auerbach, $134,076 to Mr. Eyler and $161,141 to Dr. Bryce, which represents approximately 50%, 40%, and 40%, respectively, of the executive’s annual base salary, and for Messrs. Auerbach and Eyler, and Dr. Bryce, prior to the 2015 increases.
Name | 2023 Bonus ($) | Actual Award Paid % of Target Award ($) | ||||||
Alan H. Auerbach | 407,991 | 71.50 | % | |||||
Maximo F. Nougues | 181,979 | 88.25 | % | |||||
Alvin Wong, Pharm.D. | 174,545 | 83.25 | % | |||||
Jeff J. Ludwig | 162,863 | 68.25 | % | |||||
Douglas Hunt, B.Sc (Hons) | 151,093 | 88.25 | % | |||||
Signing Bonus. In connection with entering into his employment offer letter, we paid to Mr. Lo a one-time signing bonus equal to $1,051,249. This bonus will not be earned in full until the fourth anniversary of Mr. Lo’s hire date, and is repayable, in part or in full, if his employment is terminated either by the Company for cause or by Mr. Lo for any reason, in either case prior to the fourth anniversary of his hire date.
Long-Term Equity Awards
The goalsgoal of our long-term, equity-based incentive awards areis to align the interests of our Named Executive Officers with the interests of our stockholders. Because vesting is based on continued service, our equity-based incentivesawards also encourage the retention of our Named Executive Officers during the award vesting period. In determining the size of the long-term equity incentives to be awarded to our Named Executive Officers, we take into account a number of factors, such as thecorporate performance, peer group practices as provided by Compensia, relative job scope, the value of existing stockholdings in the Company and long-term incentive awards, and individual performance history, prior financial contributions to us and the size of prior grants.history.
To reward and retain our Named Executive Officers in a manner that aligns their interests with stockholders’ interests, we have historically used stock options as the primary incentive vehicle for long-term compensation. Because employees realize value from stock options only if our stock price increases relative to the stock option’s exercise price, we believe stock options provide meaningful incentives to achieve increases in the value of our stock over time. Additionally, since 2016, we have granted our executives RSUs in addition to stock option grants, as they serve as an important retention tool.
We generally use stock optionshistorically have used equity awards to compensate our Named Executive Officers, both in the form of initial grants in connection with the commencement of employment and additional or “refresher”annual grants. We have not established a formula or program for determining the size of any equity award, including any annual refresher grants, and ourThe Compensation Committee retains discretion to make stock optionequity awards to employees at any time, including in connection with the promotion of an employee, to reward an employee, for retention purposes or in other circumstances recommended by management.
The exercise priceCompensation Committee, when determining the 2023 aggregate dollar-denominated values of each stock option grant is at or aboveequity awards for our Named Executive Officers, aligned the fair market valuetargeted aggregate dollar-denominated values with the 50th percentile of the long-term equity incentive values of our common2023 peer group, as modified by the Company’s achievement of the following 2022 fiscal year corporate objectives: revenue, cashflow, and closure of summit sites under our 2022 short-term incentive compensation program, which the Compensation Committee determined had been attained at 158.4%. In approving these increases, the Compensation Committee also reviewed and considered 2023 peer group benchmarking data that reflected the prior year’s equity grant values for our NEOs being significantly below market median, the recommendations of our CEO for an adjustment in grant value for the other NEOs, individual performance by each of the NEOs, and the limitation imposed by the shares available for grant under our 2011 Plan. These additional considerations resulted in 2023 equity grant values for each executive at 149% of peer group median for their position. Additionally, due to stock price volatility and the limit on the number of shares available in the 2011 Plan, the Compensation Committee estimated the grant date for which we usefair values of the closing price of our common stock2023 equity awards based on the applicable accounting rules and an assumed closing share price on the day of grant date. Stock option(February 10, 2023) of $4.39 per share and a Black-Scholes value per share of $3.29 and, based on this calculation, approved the number of stock options and RSUs to be granted to our Named Executive Officers in 2023.
The 2023 equity awards typicallywere granted in the form of stock options and RSUs, with each representing approximately 50% of the aggregate value of the award.
Name | 2022 Aggregate | 2023 Aggregate | ||||||
Alan H. Auerbach | 788,197 | 1,640,151 | ||||||
Maximo F. Nougues | 290,287 | 515,624 | ||||||
Alvin Wong, Pharm.D. | 209,545 | 515,624 | ||||||
Jeff J. Ludwig | 182,631 | 589,284 | ||||||
Douglas Hunt, B.Sc (Hons) | 105,734 | 423,548 | ||||||
As with the 2022 equity awards, the equity awards granted in February 2023 vest over a three-yeartwo-year period, in six month increments, as follows (subject to continued service, or for Mr. Auerbach, continued employment, through the applicable vesting date): one-thirdone-fourth (1/4th) of the shares underlyingRSUs on each of July 1, 2023, January 1, 2024, July 1, 2024 and January 1, 2025. The Compensation Committee believes that the option vest on the first anniversaryshorter vesting period in six-month increments over two years, instead of the vesting commencement date, and the remainder of the shares underlying the option vest in equal monthly installmentsannual increments over the following 24 months. However, in connection with entering into an employment offer letter with Mr. Lo, we granted him an option covering 150,000 shares that vests over a four-year period, with the 25% vesting on the first anniversary of his employment commencement date and the remainder vesting in substantially equal monthly installments thereafter, subjectfour years, continues to his continued employment. We believe these vesting schedules appropriately encourage continued service or employment, as applicable, with the Company while allowing our executives to realize compensation in line with the value they have created for our stockholders.
During 2015,2023, we made the following grants ofannual stock optionsoption grants to our Named Executive Officers. The options vest in accordance withOfficers, representing approximately 50% of the vesting schedules described above.aggregate equity award value:
2023 Stock | ||||
Options | ||||
Name | (# of Shares) | |||
Alan H. Auerbach | ||||
| ||||
| ||||
| ||||
Douglas Hunt, B.Sc (Hons) | 66,864 | |||
During 2023, we made the following annual time-based RSU grants to our Named Executive Officers, representing approximately 50% of the aggregate award value:
2023 RSUs | ||||
Name | (# of Units) | |||
Alan H. Auerbach | 186,403 | |||
Maximo F. Nougues | 58,601 | |||
Alvin Wong, Pharm.D. | 58,601 | |||
Jeff J. Ludwig | 66,972 | |||
Douglas Hunt, B.Sc (Hons) | 48,136 | |||
For 2024, consistent with the Compensation Committee’s 2023 grant methodology, long-term equity award values for our Chief Executive Officer were granted based primarily on 2023 median peer group equity grant values for CEOs, multiplied by 71.5%, which was the Company’s attainment against target under the 2023 annual short-term incentive program. There was a significant increase in median peer group CEO equity grant values for 2023. As a result of this methodology, 2024 long-term equity grant values for our Chief Executive Officer were slightly higher than 2023 equity grant values. The Compensation Committee used the same methodology to inform the 2024 grant values for the other Named Executive Officers, with revisions to these amounts made to reflect the Compensation Committee’s assessment of the Named Executive Officers’ individual performance during fiscal 2023, with Messrs. Nougues and Hunt receiving higher values and Messrs. Wong and Ludwig receiving lower values, than derived from the methodology.
2021 Auerbach Warrant
In accordance with SEC rules, Mr. Auerbach’s compensation as reported in the Summary Compensation Table of our Chief Executive Officer’s substantial rolesproxy statement includes the grant date fair values of equity awards granted in the applicable year, as well as, for 2021, the incremental fair value of $13.7 million related to an amendment to extend the term of Mr. Auerbach’s warrant (the “Auerbach Warrant”), which was approved by stockholders at the 2021 annual meeting (the “Warrant Amendment”). However, the actual delivered or realized value of these awards may differ substantially from the Summary Compensation Table reported values, as the exercise price of the warrant is $16.00 per share. In addition, due to the incremental fair value reported in 2021 with respect to the Warrant Amendment, Mr. Auerbach’s 2021 is significantly higher than his regular annual compensation reflected for 2022 and responsibilities within2023.
The Auerbach Warrant was issued on October 4, 2011 by Puma Biotechnology, Inc., a privately-held Delaware corporation just prior to it being acquired by the Company in October 2011 (the “Merger”). The Company assumed the Auerbach Warrant in the Merger, and it became exercisable in full upon the closing of the Company’s public offering of our Compensation Committee determined itcommon stock on October 24, 2012 (the “2012 Public Offering”). At that time, the number of shares subject to the Auerbach Warrant was appropriateset at 2,116,250 shares, based on the number of shares sufficient to maintain beneficial ownership of 20% of our outstanding shares of common stock as of the closing of our 2012 Public Offering, and the exercise price was set at the 2012 Public Offering price per share of $16.00.
The Auerbach Warrant originally was granted in recognition of Mr. Auerbach’s many contributions to the Company with the objectives of allowing him the ability to maintain beneficial ownership of at least 20% of our outstanding common stock, encouraging full investment of his time and energy to further our best interests, and providing incentives for stock appreciation. If the Warrant Amendment had not been approved, then the Auerbach Warrant would have expired pursuant to its original terms, and would have been forfeited by Mr. Auerbach in full. When approving the Warrant Amendment in April 2021, the Board considered the importance for Mr. Auerbach to receivehave the right to maintain a larger stock option grant than thosesignificant ownership interest in the Company to best align his interests with that of our other Named Executive Officers. In addition,stockholders and long-term value creation. The Board also believed that the size of Mr. Lo’s 2015 option grant was larger as it was negotiatedAuerbach Warrant provides a valuable incentive to increase our stock price.
Although the Board does not view the Auerbach Warrant as part of Mr. Auerbach’s standard compensation program for 2021, SEC rules require the Company to disclose the incremental fair value associated with the Warrant Amendment, as determined under applicable accounting standards. In accordance with ASC 718, the extension of the term of the Auerbach Warrant resulted in a generally non-cash, stock-based compensation expense of $13.7 million, as reported under the “Option Awards” column for the 2021 fiscal year of the Summary Compensation Table. Mr. Auerbach’s 2021 equity compensation excluding the amount attributed to the Auerbach Warrant was $3.6 million, which represented a small increase from his initial hiringequity compensation package.of $3.15 million in 2020.
Severance and Change in Control Arrangements
Mr. Auerbach’s employment agreement and Mr. Lo’sMessrs. Nougues’ and Ludwig’s employment agreementsletters provide that the executive is eligible to receive severance payments and benefits upon a qualifying involuntary termination of employment, including in
connection with a change in control of our company.Company (as opposed to solely upon a “single-trigger” change in control). We believe that these protections serve to encourage continued attention and dedication to duties without distraction arising from the possibility of a change in control, and provide the business with a smooth transition in the event of such a termination of employment in connection with a transaction. These severance and change in control arrangements are designed to retain certain of our executives in these key positions as we compete for talented executives in the marketplace where such protections are commonly offered. For a detailed description of the severance provisions contained in Mr. Auerbach’s employment agreement and Mr. Lo’sMessrs. Nougues’ and Ludwig’s employment agreements,letters, see “Potential Payments Upon Termination or Change in Control” below.
In addition, Mr. Lo’s employment agreement provides he is eligible to receive severance payments and benefits upon a qualifying termination, including in connection with a change in control of our company. For a detailed description of the severance provisions contained in Mr. Lo’s employment agreement, see “Potential Payments Upon Termination or Change in Control” below.
Other Elements of Compensation and Perquisites
We provide our executive officers, including our Named Executive Officers, with long-term disability and long-term care insurance, and we reimburse for physical exams. All of our full-time employees in the United States, including our Named Executive Officers, are eligible to participate in our 401(k) plan. Pursuant to our 401(k) plan, employees may elect to reduce their current compensation by up to the statutorily prescribed annual limit, which was $18,000$22,500 in 2015,2023, or $30,000 for employees age 50 and older, and to have the amount of this reduction contributed to our 401(k) plan. In addition, all of our full-time employees, including our Named Executive Officers, are eligible to participate in our health and welfare plans.
Prohibition on Hedging and Pledging
We maintain an Insider Trading Compliance Policy that prohibits hedging by our officers, directors and employees, pledging of Company stock as collateral to secure loans, margin purchases of the Company’s stock, short sales of the Company’s stock, and any transactions in puts, calls or other derivative securities involving the Company’s stock.
Clawback Policy
We believe in maintaining best practices for our executive compensation program, and as part of that our Board in 2023 adopted a compensation recovery policy that requires the recovery of certain erroneously paid incentive compensation received by our Section 16 officers on or after October 2, 2023, in the event the Company is required to prepare a financial restatement, as defined, as required by new SEC rules and NASDAQ listing standards implemented pursuant to the Dodd-Frank Wall Street Reform and Consumer Protection Act (the “Dodd-Frank Act”), and which can be recovered from time-vesting or performance-vesting equity compensation (in addition to other forms of incentive compensation).
Executive Stock Ownership Guidelines
We maintain stock ownership guidelines covering our executive officers, including our Named Executive Officers. We believe that linking a significant portion of an officer’s current and potential future net worth to our success, as reflected in our share price, helps to ensure that officers have a stake similar to that of our stockholders. Stock ownership guidelines also encourage long-term management of the Company for the benefit of its stockholders.
These guidelines require the covered individual to own an amount of our common stock with an aggregate market value equal to a specified multiple of their base salary. Each covered individual is expected to satisfy the applicable ownership requirement generally within five years after first becoming subject to the guidelines. The table below reflects the current ownership guidelines for executives:
Position | MultipleofBaseSalary |
Chief Executive Officer | 3x |
Executive Officers (other than the Chief Executive Officer) | 1x |
The types of ownership arrangements counted towards the guidelines are: shares of our common stock, whether held individually, jointly, or in trust with or for the benefit of an immediate family member, shares held by a corporate entity in which the individual holds voting or disposal power over such shares, or unvested restricted stock unit awards that vest solely based on the passage of time. As of December 31, 2023, each Named Executive Officer had met these ownership requirements.
Tax and Accounting Considerations
Section 162(m) of the Code
Section 162(m) of the Code disallows a tax deduction for any publicly held corporation for individual compensation exceeding $1.0 million in any taxable year for the group of individuals generally comprised of its named executive officers, other than its chief financial officer, unless compensation is performance-based. Where reasonably practicable and to the extent that the Section 162(m) deduction disallowance becomes applicable to our company, our Compensation Committee may seek to qualify the variable compensation paid to our executive officers for an exemption from the deductibility limitations. As such, in approving the amount and form of compensation for our executive officers in the future, our Compensation Committee has and will continue to consider all elements of the cost to our company of providing such compensation, including the potential impact of Section 162(m) of the Code. However, our Compensation Committee may, in its judgment, authorize compensation payments that do not comply with the exemptions in Section 162(m) of the Code when it believes that such payments are appropriate to attract and retain executive talent.
Section 280G of the Code
Section 280G of the Internal Revenue Code of 1986, as amended (the “Code”) disallows a tax deduction with respect to excess parachute payments to certain executives of companies whichthat undergo a change in control. In addition, Section 4999 of the Code imposes a 20% excise tax on the individual with respect to the excess parachute payment. Parachute payments are compensation linked to or triggered by a change in control and may include, but are not limited to, bonus payments, severance payments, certain fringe benefits, and payments and acceleration of vesting from long-term incentive plans including stock options and other equity-based compensation. Excess parachute payments are parachute payments that exceed a threshold determined under Section 280G of the Code based on the executive’s prior compensation.
Under his employment agreement, Mr. Auerbach may be entitled to a gross-up payment that will make him whole in the event that any parachute payment excise taxes are imposed. We provide this protection to Mr. Auerbach to help ensure that he will be properly incentivized in the event of a potential change in control of the Company to maximize stockholder value in a transaction without concern for potential consequences of the transaction to him.
In approving the compensation arrangements for our named executive officersNamed Executive Officers in the future, ourthe Compensation Committee will consider all elements of the cost to the Company of providing such compensation, including the potential impact of Section 280G of the Code. However, ourthe Compensation Committee may, in its judgment, authorize compensation arrangements that could give rise to loss of deductibility under Section 280G of the Code and the imposition of excise taxes under Section 4999 of the Code when it believes that such arrangements are appropriate to attract and retain executive talent.
Under his employment agreement, Mr. Auerbach may be entitled to a gross-up payment that will make him whole in the event that any parachute payment excise taxes are imposed on him in excess of a certain threshold. We provide this protection to Mr. Auerbach to help ensure that he will be properly incentivized in the event of a potential change in control of the Company to maximize shareholder value in a transaction without concern for potential consequences of the transaction to him.
Accounting Standards
ASC Topic 718,Compensation – Stock Compensation (“(“ASC Topic 718”) requires us to recognize an expense for the fair value of equity-based compensation awards. Grants of stock options, restricted stock, performance shares and restricted stock units (“RSUs”)RSUs under our equity incentive award plans are accounted for under ASC Topic 718. Our
The Compensation Committee regularly considers the accounting implications of significant compensation decisions, especially in connection with decisions that relate to our equity incentive award plans and programs. As accounting standards change, we may revise certain programs to appropriately align accounting expenses of our equity awards with our overall executive compensation philosophy and objectives.
The following table sets forth information regarding the compensation earned by our Named Executive Officers for the years ended December 31, 2015, 20142023, 2022 and 2013.2021.
Name and Principal Position | Year | Salary ($) | Bonus ($)(1) | Option Awards ($)(2) | All Other Compensation ($) | Total ($) | ||||||||||||||||||
Alan H. Auerbach | 2015 | 651,000 | 315,000 | 6,838,394 | 11,260 | (3) | 7,815,654 | |||||||||||||||||
President and Chief Executive Officer | 2014 | 610,000 | 300,000 | 16,876,576 | 11,030 | 17,797,606 | ||||||||||||||||||
| 2013 | 546,667 | 260,000 | 4,555,285 | 10,620 | 5,372,572 | |||||||||||||||||||
Charles R. Eyler | 2015 | 344,472 | 134,076 | 1,417,795 | 17,458 | (3) | 1,913,801 | |||||||||||||||||
Senior Vice President, Finance and Administration | 2014 | 304,336 | 117,610 | 4,061,879 | 15,734 | 4,499,559 | ||||||||||||||||||
| 2013 | 282,900 | 83,475 | 901,228 | 15,817 | 1,283,420 | |||||||||||||||||||
Richard P. Bryce, MBChB, MRCGP and MFPM | 2015 | 371,782 | 161,141 | 1,593,077 | 12,664 | (3) | 2,138,664 | |||||||||||||||||
Senior Vice President, Clinical Research and Development | 2014 | 342,692 | 133,126 | 1,448,393 | 12,206 | 1,936,417 | ||||||||||||||||||
| 2013 | 322,724 | 110,250 | 1,398,532 | 18,584 | 1,850,086 | |||||||||||||||||||
Steven Lo | 2015 | 133,766 | 1,051,249 | 7,947,658 | 247 | (3) | 9,132,920 | |||||||||||||||||
Chief Commercial Officer(4) | ||||||||||||||||||||||||
Name and Principal Position | Year | Salary ($) | Bonus ($) (1) | Stock Awards ($) (2) | Option Awards ($) (3) | Non-Equity Incentive Plan Compensation ($) (4) | All Other Compensation ($) (5) | Total ($) | |||||||||||||||||||||||
Alan H. Auerbach | 2023 | 877,871 | — | 818,309 | 821,842 | 407,991 | 18,735 | 2,944,748 | |||||||||||||||||||||||
President and Chief Executive Officer | 2022 | 852,302 | — | 393,360 | 394,837 | 877,530 | 14,732 | 2,532,761 | |||||||||||||||||||||||
2021 | 827,478 | — | 1,802,411 | 15,540,659 | 268,930 | 14,132 | 18,453,610 | (6) | |||||||||||||||||||||||
Maximo F. Nougues | 2023 | 515,521 | 108,259 | 257,258 | 258,366 | 73,720 | 18,702 | 1,231,826 | |||||||||||||||||||||||
Chief Financial Officer | 2022 | 495,693 | 114,009 | 144,870 | 145,417 | 157,036 | 14,732 | 1,071,757 | |||||||||||||||||||||||
2021 | 472,089 | 94,418 | 668,252 | 668,160 | 47,209 | 14,132 | 1,964,260 | ||||||||||||||||||||||||
Alvin Wong, Pharm.D. | 2023 | 524,160 | 99,590 | 257,258 | 258,366 | 74,955 | 15,535 | 1,229,864 | |||||||||||||||||||||||
Chief Scientific Officer | 2022 | 504,000 | 115,920 | 104,575 | 104,970 | 159,667 | 30,104 | 1,019,237 | |||||||||||||||||||||||
2021 | 459,364 | 96,000 | 84,692 | 651,954 | 48,000 | 22,424 | 1,362,434 | ||||||||||||||||||||||||
Jeff J. Ludwig | 2023 | 596,568 | 77,554 | 294,007 | 295,277 | 85,309 | 33,134 | 1,381,848 | |||||||||||||||||||||||
Chief Commercial Officer | 2022 | 573,623 | 131,933 | 91,145 | 91,486 | 181,724 | 29,572 | 1,099,483 | |||||||||||||||||||||||
2021 | 562,375 | 89,980 | 433,549 | 433,485 | 56,238 | 26,416 | 1,602,043 | ||||||||||||||||||||||||
Douglas Hunt, B.Sc (Hons) | 2023 | 428,026 | 89,885 | 211,317 | 212,231 | 61,208 | 18,886 | 1,021,553 | |||||||||||||||||||||||
Chief Regulatory Affairs, Medical Affairs, | 2022 | 398,164 | 91,578 | 52,768 | 52,966 | 126,138 | 16,767 | 738,381 | |||||||||||||||||||||||
Pharmacovigilance, and Law Officer | 2021 | 379,204 | 75,841 | 237,479 | 237,443 | 37,920 | 16,075 | 983,962 | |||||||||||||||||||||||
(1) | Reflects the discretionary |
(2) | Represents the grant date fair values of |
(3) | Represents the grant date fair values of stock options granted during the applicable year determined in accordance with ASC 718. For a discussion of valuation assumptions for the stock option grants, see Note 10 to our 2023 Consolidated Financial Statements included in our Annual Report on Form 10-K for the year ended December 31, 2023 filed with the SEC on February 29, 2024. There can be no assurance that awards will vest or will be exercised (if they are not exercised, no value will be realized by the individual), or that the value upon exercise will approximate the aggregate grant date fair value determined under ASC 718. With respect to Mr. Auerbach, in accordance with the requirements of ASC 718 and SEC rules, the reported amount for 2021 also includes $13,738,509 in incremental fair value related to the amendment to extend the term of the Auerbach Warrant which was approved by our stockholders at the 2021 annual meeting. Excluding the incremental fair value attributed to the Auerbach Warrant extension, Mr. Auerbach’s 2021 annual Stock Awards compensation was $1,802,150. |
(4) | Reflects annual short-term incentive compensation payments for the applicable year earned under the Corporate Performance Portion of our short-term incentive compensation program. |
(5) | For |
(6) | The total compensation reported for Mr. |
Grants of Plan-Based Awards in 20152023
The following table sets forth information regarding grants of plan-based awards made to our Named Executive Officers during the year ended December 31, 2015.2023.
Name | Grant Date | All Other Option Awards: Number of Shares Underlying Options (#) | Exercise or Base Price of Option Awards Per Share ($) | Grant Date Fair Value of Option Awards ($)(1) | ||||||||||||
Alan H. Auerbach | 10/7/2015 | 150,000 | 76.54 | 6,838,394 | ||||||||||||
Charles R. Eyler | 10/8/2015 | 31,500 | 75.52 | 1,417,795 | ||||||||||||
Richard P. Bryce, MBChB, MRCGP, MFPM | 8/3/2015 | 31,500 | 87.88 | 1,593,077 | ||||||||||||
Steven Lo | 9/8/2015 | 150,000 | 95.22 | 7,947,658 | ||||||||||||
Estimated Future Payouts Under Non-Equity Incentive Plan Awards | Stock Awards: Number of | Option Awards: Number of Shares | Exercise or Base Price of Option | Grant Date Fair Value of Stock | |||||||||||||||||||||||||
Name | Grant Date | Threshold ($) | Target ($) | Maximum ($) | Shares or Units (#) (2) | Underlying Options (#) (3) | Awards Per Share ($) | and Option Awards ($)(4) | |||||||||||||||||||||
Alan H. Auerbach | 2/10/2023 | — | — | — | — | 258,924 | 4.39 | 821,842 | |||||||||||||||||||||
2/10/2023 | — | — | — | 186,403 | — | — | 818,309 | ||||||||||||||||||||||
| — | — | — | — | — | — | — | |||||||||||||||||||||||
| (1 | ) | 570,616 | 1,141,232 | — | — | — | — | ||||||||||||||||||||||
Maximo F. Nougues | 2/10/2023 | — | — | — | — | 81,399 | 4.39 | 258,366 | |||||||||||||||||||||
2/10/2023 | — | — | — | 58,601 | — | — | 257,258 | ||||||||||||||||||||||
| (1 | ) | 103,104 | 206,208 | — | — | — | — | ||||||||||||||||||||||
Alvin Wong, Pharm.D. | 2/10/2023 | — | — | — | — | 81,399 | 4.39 | 258,366 | |||||||||||||||||||||
2/10/2023 | — | — | — | 58,601 | — | — | 257,258 | ||||||||||||||||||||||
| (1 | ) | 104,832 | 209,664 | — | — | — | — | ||||||||||||||||||||||
Jeff J. Ludwig | 2/10/2023 | — | — | — | — | 93,028 | 4.39 | 295,277 | |||||||||||||||||||||
2/10/2023 | — | — | — | 66,972 | — | — | 294,007 | ||||||||||||||||||||||
| (1 | ) | 119,314 | 238,627 | — | — | — | — | ||||||||||||||||||||||
Douglas Hunt, B.Sc (Hons) | 2/10/2023 | — | — | — | — | 66,864 | 4.39 | 212,231 | |||||||||||||||||||||
2/10/2023 | — | — | — | 48,136 | — | — | 211,317 | ||||||||||||||||||||||
| (1 | ) | 85,605 | 171,210 | — | — | — | — | ||||||||||||||||||||||
(1) | The amounts in these columns represent the range of potential payouts under the Corporate Performance Portion of our 2023 annual short-term incentive compensation program based on achievement of pre-established goals. For Mr. Auerbach, 100% of his entire target short-term incentive compensation opportunity was based on the Corporate Performance Portion of our short-term incentive compensation program. For our other Named Executive Officers, 50% of each executive’s entire target short-term incentive compensation opportunity was based on the Corporate Performance Portion of our short-term incentive compensation program, and 50% is based on Compensation Committee discretion based on individual performance and thus is not reflected in this table. Achievement at or below threshold results in no payout, with straight line interpolation between threshold and target (0-100%) and between target and threshold (100-200%). |
(2) | Amounts reflect the number of RSUs granted in 2023 with one-fourth (1/4) of the RSUs vesting on each of July 1, 2023, January 1, 2024, July 1, 2024 and January 1, 2025, subject to continued service, or for Mr. Auerbach, continued employment, through the applicable vesting date. |
(3) | Amounts reflect the number of stock options granted in 2023 with one-fourth (1/4) of the shares underlying the option vesting on each of July 1, 2023, January 1, 2024, July 1, 2024 and January 1, 2025, subject to continued service, or for Mr. Auerbach, continued employment, through the applicable vesting date. |
(4) | Represents the grant date fair values of |
Narrative Disclosure to Summary Compensation Table and Grants of Plan-Based Awards Table
Offer Letters and Employment Agreements with Our Named Executive Officers
President and Chief Executive Officer – Alan H. Auerbach
On January 19, 2012, we entered into an employment agreement with Alan H. Auerbach, our President and Chief Executive Officer. The employment agreement governs the terms of Mr. Auerbach’s employment with us and expired onprovided for an initial term through September 1, 2014, but is subject to automatic one-year renewal terms unless earlier terminated or either we or Mr. Auerbach givesgive written notice of termination 60 days prior to the end of the renewed term. The employment agreement also provides that Mr. Auerbach will be nominated for election to our Board if the term of his directorship expires during the term of the employment agreement.
Pursuant to the employment agreement, Mr. Auerbach will receive an annual base salary which(which was initially set at $470,000,$877,871 in 2023), and he is eligible to receive an annual discretionary bonusaward under our short-term incentive compensation program (which in an amount up to 50%2023 was targeted at 65% of his annual base salary (pro-ratedsalary), pro-rated for any partial year service),service, each subject to possible increase in connection with our annual review process. Mr. Auerbach is also eligible under the employment agreement to participate in all benefits offered to our senior executives. In connection with entering into the employment agreement, Mr. Auerbach also received an option to purchase 200,000 shares of our common stock in February 2012, which vested in full in 2016.
For a discussion of the payments and other benefits to which Mr. Auerbach is entitled under his employment agreement in the event of certain qualifying terminations, including certain terminations in connection with a change in control of us, see “Potential Payments Upon a Termination or Change in Control” below.
Mr. Auerbach’s employment agreement contains customary confidentiality and assignment of inventions provisions that survive the termination of the employment agreement for an indefinite period. The employment agreement also contains non-solicitation and non-disparagement provisions extending until 18 months following the termination of his employment with us.
Other Named Executive Officers – Charles R. Eyler, Richard P. Bryce, MBChB, MRCGP, MFPM Maximo F. Nougues, Jeff J. Ludwig, Douglas Hunt, and Steven LoAlvin Wong
We have entered into employment letter agreements with each of theour other Named Executive Officers, listed in the table below on the date set forth next to such officer’s name. Messrs. Nougues, Ludwig, Hunt and Dr. Wong.
These Named Executive Officers are at-will employees. The
table below also sets forthemployees and each officer’semployment letter provides for an initial base salary and target annual bonus opportunity underexpressed as a percentage of their salary, which are subject to increase at the letter agreements. Pursuant todiscretion of the agreements, these executives are eligible to receive aCompensation Committee, discretionary annual performance bonusbonuses subject to the attainment of performance criteria established and evaluated by us.
Name | Offer Letter Date | Initial Base Salary ($) | Target Annual Bonus (% of Base Salary) | |||||||||
Charles R. Eyler | October 21, 2011 | $ | 265,000 | 30 | % | |||||||
Richard P. Bryce, MBChB, MRCGP, MFPM | May 2, 2012 | $ | 315,000 | 35 | % | |||||||
Steven Lo | August 17, 2015 | $ | 425,000 | 40 | % | |||||||
us, and an initial option grant that vests over a three-year period. Each of Mr. Eyler, Dr. Bryce and Mr. Loemployment letter also provides that the executive is also eligible to participate in all health, welfare, savings and retirement plans, practices, policies and programs maintained or sponsored by us from time to time for the benefit of similarly situated employees. In addition, Mr. Ludwig’s employment letter agreement provides that Mr. Ludwig will be eligible to receive one or more equity awards based on the achievement of certain sales milestones. No equity awards were granted in accordance with these provisions during 2023.
Additionally, the employment letters for Messrs. Nougues and Ludwig include certain payments and other benefits to which the executive is entitled in the event of certain qualifying terminations, including certain terminations in connection with entering intoa change in control of us. Please see “Potential Payments Upon a Termination or Change in Control” below for a description of these letter agreements, we granted Mr. Eyler an option to purchase 90,000 shares of our common stock, Dr. Bryce an option to purchase 105,000 shares of our common stock and Mr. Lo an option to purchase 150,000 shares of our common stock. The initial options to Mr. Eyler and Dr. Bryce are vested in full.provisions.
The letter agreementsemployment letters also contain a customary non-solicitation provision and, in connection with their entry into the offeremployment letters, each of the Named Executive Officers listed in the table aboveMessrs. Nougues, Ludwig, Hunt and Dr. Wong entered into our standard proprietary information and inventions agreement.
Outstanding Equity Awards at Fiscal Year End
The following table sets forth summary information regarding the outstanding equity awards held by our Named Executive Officers at December 31, 2015.2023. Except as indicated below, stock awards and options were granted pursuant to our 2011 Plan.
| Option Awards | ||||||||||||||||
Name | Number of Securities Underlying Unexercised Options (#) Exercisable | Number of Securities Underlying Unexercised Options (#) Unexercisable | Option Exercise Price ($) | Option Expiration Date | ||||||||||||
Alan H. Auerbach(1) | 200,000 | — | (2) | 3.75 | 2/13/2022 | |||||||||||
| 150,000 | — | (3) | 19.34 | 12/17/2022 | ||||||||||||
| 108,333 | 41,667 | (4) | 44.08 | 10/25/2023 | ||||||||||||
| 62,499 | 87,501 | (5) | 195.33 | 12/15/2024 | ||||||||||||
| — | 150,000 | (6) | 76.54 | 10/6/2025 | ||||||||||||
Charles R. Eyler | 90,000 | — | (7) | 3.75 | 2/13/2022 | |||||||||||
| 22,500 | — | (8) | 19.34 | 12/17/2022 | ||||||||||||
| 21,874 | 9,626 | (9) | 41.50 | 11/18/2023 | ||||||||||||
| 13,124 | 18,376 | (10) | 223.32 | 11/19/2024 | ||||||||||||
| — | 31,500 | (11) | 75.52 | 10/7/2025 | ||||||||||||
Richard P. Bryce, MBChB, MRCGP, MFPM | 102,000 | — | (12) | 11.30 | 6/1/2022 | |||||||||||
| 29,604 | 7,146 | (13) | 55.20 | 7/9/2023 | ||||||||||||
| 14,874 | 16,626 | (14) | 65.66 | 7/9/2024 | ||||||||||||
| — | 31,500 | (15) | 87.88 | 8/2/2025 | ||||||||||||
Steven Lo | — | 150,000 | (16) | 95.22 | 9/7/2025 | |||||||||||
Option Awards | Stock Awards | |||||||||||
Name | Number of | Number of | Option | Option | Number of | Market Value | ||||||
Alan H. Auerbach | 2,116,250 | (2) | — | 16.00 | 10/4/2026 | — | — | |||||
150,000 | — | 195.33 | 12/12/2024 | — | — | |||||||
150,000 | — | 76.54 | 10/4/2025 | — | — | |||||||
66,667 | — | 35.61 | 5/22/2026 | — | — | |||||||
70,000 | — | 37.35 | 2/16/2027 | — | — | |||||||
56,940 | — | 60.85 | 2/11/2028 | — | — | |||||||
62,634 | — | 27.76 | 2/6/2029 | — | — | |||||||
161,649 | — | 12.34 | 2/11/2030 | — | — | |||||||
175,800 | 35,144 | (3) | 12.02 | 2/17/2031 | 24,982 | (4) | 108,172 | |||||
180,882 | 60,294 | (5) | 2.33 | 3/3/2032 | 42,206 | (6) | 182,752 | |||||
64,731 | 194,193 | (7) | 4.39 | 2/10/2033 | 139,803 | (8) | 605,347 | |||||
Maximo F. Nougues | 90,000 | — | 23.37 | 11/11/2028 | — | — | ||||||
15,567 | — | 12.34 | 2/11/2030 | — | — | |||||||
65,179 | 13,030 | (3) | 12.02 | 2/17/2031 | 9,263 | (4) | 40,109 | |||||
66,618 | 22,206 | (5) | 2.33 | 3/3/2032 | 15,544 | (6) | 67,306 | |||||
20,349 | 61,050 | (7) | 4.39 | 2/10/2033 | 43,951 | (8) | 190,308 | |||||
Alvin Wong, Pharm.D. | 10,500 | — | 112.80 | 3/14/2024 | — | — | ||||||
10,500 | — | 217.38 | 3/23/2025 | — | — | |||||||
7,035 | — | 73.57 | 11/30/2025 | — | — | |||||||
10,500 | — | 39.50 | 12/1/2026 | — | — | |||||||
75,000 | 15,000 | (9) | 10.21 | 6/17/2031 | — | — | ||||||
48,088 | 16,030 | (5) | 2.33 | 3/3/2032 | 11,221 | (6) | 48,587 | |||||
20,349 | 61,050 | (7) | 4.39 | 2/10/2033 | 43,951 | (8) | 190,308 | |||||
Jeff J. Ludwig | 320,000 | — | 7.82 | 3/20/2030 | — | — | ||||||
42,286 | 8,454 | (3) | 12.02 | 2/17/2031 | 6,010 | (4) | 26,023 | |||||
41,911 | 13,971 | (5) | 2.33 | 3/3/2032 | 9,780 | (6) | 42,347 | |||||
23,257 | 69,771 | (7) | 4.39 | 2/10/2033 | 50,229 | (8) | 217,492 | |||||
Douglas Hunt, B.Sc (Hons) | 90,000 | — | 90.85 | 1/16/2028 | — | — | ||||||
15,400 | — | 27.76 | 2/6/2029 | — | — | |||||||
54,842 | — | 12.34 | 2/11/2030 | — | — | |||||||
23,162 | 4,631 | (3) | 12.02 | 2/17/2031 | 3,292 | (4) | 14,254 | |||||
24,264 | 8,089 | (5) | 2.33 | 3/3/2032 | 5,662 | (6) | 24,516 | |||||
16,716 | 50,148 | (7) | 4.39 | 2/10/2033 | 36,102 | (8) | 156,322 | |||||
(1) | Market value is determined based on the |
(2) | Mr. Auerbach holds a warrant that was issued in 2011 and is exercisable until October |
(3) |
| One-third of the granted option | |
(4) | One-third of the granted RSU award vested on the first anniversary of the vesting commencement date of January 1, 2021, one-sixth vested on July 1, 2022, January 1, 2023, and July 1, 2023, with this last unvested increment vesting on January 1, 2024, subject to continued service, or for Mr. Auerbach, continued employment, through the applicable vesting date. In addition, for Messrs. Auerbach, Nougues, and Ludwig the RSUs may accelerate and vest under certain circumstances described more fully under “Potential Payments Upon a Termination or Change in Control” below. |
(5) | One-fourth of the granted option vested on July 1, 2022, January 1, 2023, and July 1, 2023, with this last unvested increment vesting on January 1, 2024, subject to continued service, or for Mr. Auerbach, continued employment, through the applicable vesting date. In addition, for Messrs. Auerbach, Nougues, and Ludwig this option may accelerate and vest under certain circumstances described more fully under “Potential Payments Upon a Termination or Change in Control” below. |
(6) | One-fourth of the |
(7) | One-fourth of the granted option vested on July 1, 2023, and these remaining unvested options will vest in one-fourth increments on each of January 1, 2024, July 1, 2024 and January 1, 2025, subject to continued service, or for Mr. Auerbach, continued employment, through the applicable vesting date. In addition, for Messrs. Auerbach, Nougues, and Ludwig this option may accelerate and vest under certain circumstances described more fully under “Potential Payments Upon a Termination or Change in Control” below. |
(8) | One-fourth of the |
(9) |
One-third of the granted option |
Option ExercisesOptions Exercised and Stock Vested
The following table shows the number of shares of common stock acquired by each named executive officerNamed Executive Officer during 20152023 upon the exercisevesting of options held by each named executive officerRSUs during 2015.2023. None of our named executive officers held any outstandingNamed Executive Officers exercised stock awards in 2015.options during 2023.
| Option Awards | Stock Awards | |||||||||||||||
Name | Number of Shares Acquired on Exercise (#) | Value Realized on Exercise ($)(1) | Number of Shares Acquired on Vesting (#) | Value Realized on Vesting ($) | ||||||||||||
Alan H. Auerbach | — | — | — | — | ||||||||||||
Charles R. Eyler | — | — | — | — | ||||||||||||
Richard P. Bryce, MBChB, MRCGP, MFPM | 3,000 | 296,184 | — | — | ||||||||||||
Steven Lo | — | — | — | — | ||||||||||||
Stock Awards | ||||||||
Number of | Value | |||||||
Shares | Realized on | |||||||
Acquired on | Vesting | |||||||
Name | Vesting (#) | ($) (1) | ||||||
Alan H. Auerbach | 184,550 | 700,984 | ||||||
Maximo F. Nougues | 64,706 | 246,084 | ||||||
Alvin Wong, Pharm.D. | 39,275 | 146,637 | ||||||
Jeff J. Ludwig | 48,327 | 181,649 | ||||||
Douglas Hunt, B.Sc (Hons) | 31,148 | 117,063 | ||||||
(1) | Represents the amounts realized based on the closing stock price |
Pension Benefits and Nonqualified Deferred Compensation
During the fiscal year ended December 31, 2015,2023, we did not have any plans in place for the payment of retirement benefits or benefits that will be paid primarily following retirement including, but not limited to, tax qualified deferred benefit plans, supplemental executive retirement plans, tax qualified deferred contribution plans and nonqualified deferred contribution plans. We do maintain a 401(k) plan for our employees, including our NEOs.
Potential Payments Upon a Termination or Change in Control
Alan H. Auerbach. On January 19, 2012, we entered into an employment agreement with Alan H. Auerbach, our President and Chief Executive Officer. Pursuant to the employment agreement, in the event Mr. Auerbach’s employment is terminated by us without “cause” or by Mr. Auerbach for “good reason” 60 days prior to, or 18 months following, a “change in control” (each as defined in the employment agreement and described below), he will be entitled to receive, in addition to any accrued but unpaid compensation and benefits:
a lump sum payment equal to two times the sum of his base salary and the maximum bonus to which he would be eligible to receive for the year in which the termination occurs;
| • | A lump sum payment equal to two times the sum of his base salary and the maximum amount payable under our annual short-term incentive compensation program to which he would be eligible to receive for the year in which the termination occurs; |
all unvested equity-based incentive awards will immediately vest on the later of the change in control and the termination date, and will remain exercisable (as applicable) for a period of up to 12 months from the date of the termination; and
| • | all unvested equity-based incentive awards will immediately vest in connection with such termination of employment on the later of the change in control and the termination date, and will remain exercisable (as applicable) for a period of up to 12 months from the date of the termination; and |
| • | up to 18 months continuation of healthcare benefits to him and his dependents. |
up to 18 months continuation of healthcare benefits to him and his dependents.
In the event a change in control occurs and an excise tax is imposed as a result of any compensatory payments made to Mr. Auerbach in connection with such change in control, we will either (i) reduce the payments made to Mr. Auerbach such that the excise tax will not be imposed or (ii) if such reduction would not result in certain circumstances,a greater net after-tax benefit to Mr. Auerbach than receiving the full amount of such payments, pay the entire (unreduced) payments and also pay or reimburse Mr. Auerbach an amount equal to any such excise tax plus any taxes resulting from such payments.
In the event Mr. Auerbach’s employment is terminated without “cause” or by Mr. Auerbach for “good reason,” in each case outside of the change in control context described above, then Mr. Auerbach will be entitled to receive, in addition to any accrued but unpaid compensation and benefits (i) an amount equal to the sum of his base salary and the maximum bonus to which he would be eligible to receive for the year in which the termination occurs, payable over a period of one year following such termination in substantially equal installments; and (ii) up to 18 months continuation of healthcare benefits to him and his dependents. benefits:
| • | An amount equal to the sum of his base salary and the maximum amount under our annual short-term cash incentive compensation program to which he would be eligible to receive for the year in which the termination occurs, payable over a period of one year following such termination in substantially equal installments; and |
| • | up to 18 months continuation of healthcare benefits to him and his dependents. |
All severance benefits are contingent upon Mr. Auerbach’s execution and non-revocation of a general release of claims in favor of us. Under the terms of Mr. Auerbach’s employment agreement:
“Cause” is generally defined as (i) the willful failure, disregard or refusal by the executive to perform his duties; (ii) any willful, intentional or grossly negligent act by the executive that injures in a material way our business or reputation; (iii) willful misconduct by the executive in respect of his duties or obligations; (iv) the executive’s commission of any felony or a misdemeanor involving moral turpitude (including entry of a nolo contendere plea to any such charge); (v) the determination by us, after a reasonable and good-faith investigation following a written allegation by another employee of us that the executive engaged in some form of harassment prohibited by law, unless the executive’s actions were specifically directed by the board; (vi) any misappropriation or embezzlement of our property; (vii) breach by the executive of his obligations with respect to confidentiality, non-solicitation and non-disparagement or of any of his representations or warranties under the employment agreement; and (viii) material breach by the executive of any other provision of the employment agreement which is not cured within a specified timeframe.
“Good reason” is generally defined as: (i) a material diminution in the executive’s base salary, excluding any reduction applicable equally to all of our executive officers following a material decline in our earnings, public image, or performance; (ii) a material diminution in the executive’s authority,
| • | “Cause” is generally defined as (i) the willful failure, disregard or refusal by the executive to perform his duties; (ii) any willful, intentional or grossly negligent act by the executive that injures in a material way our business or reputation; (iii) willful misconduct by the executive in respect of his duties or obligations; (iv) the executive’s commission of any felony or a misdemeanor involving moral turpitude (including entry of a nolo contendere plea to any such charge); (v) the determination by us, after a reasonable and good-faith investigation following a written allegation by another employee of us that the executive engaged in some form of harassment prohibited by law, unless the executive’s actions were specifically directed by the board; (vi) any misappropriation or embezzlement of our property; (vii) breach by the executive of his obligations with respect to confidentiality, non-solicitation and non-disparagement or of any of his representations or warranties under the employment agreement; and (viii) material breach by the executive of any other provision of the employment agreement which is not cured within a specified timeframe. |
| • | “Good reason” is generally defined as: (i) a material diminution in the executive’s base salary, excluding any reduction applicable equally to all of our executive officers following a material decline in our earnings, public image, or performance; (ii) a material diminution in the executive’s authority, duties or responsibilities; (iii) a change in the geographic location at which the executive must perform services to a location that is greater than 25 miles from our principal place of business as of the date of the employment agreement; (iv) a direction to the executive to take any action that violates any applicable legal or regulatory requirement; or (v) any other action or inaction that constitutes a material breach by us of our obligations under the employment agreement. |
| • | A “change in control” is generally defined as: (i) the consummation of a transaction where any persons become the beneficial owners of Company securities representing more than 50% of the total combined voting power of our securities after such acquisition; (ii) a change in the composition of the board such that during any period of two consecutive years, individuals who originally formed our board,
Pursuant to the employment letter agreements, in the event the executive’s employment is terminated by us without “cause” or by the executive for “good reason” (each as defined in the applicable employment letter and described below) he will be entitled to receive, in addition to any
All severance benefits are contingent upon the executive’s execution and non-revocation of a general release of claims in
Terms of Equity Awards. Under the terms of the 2011 Plan, if, in the event of a change in control (as defined above), The following table summarizes the payments that would have been made to 34 The following table also assumes that a successor corporation would assume or provide substitute equity awards in a change in control.
As required by Section 953(b) of the Dodd-Frank Act, and Item 402(u) of Regulation S-K, we are providing the following information regarding the ratio of the annual total compensation of the individual identified as our median paid employee to the annual total compensation of Alan H. Auerbach, our President and Chief Executive Officer (our “CEO”). We consider the pay ratio specified below to be a reasonable estimate, calculated in a manner that is intended to be consistent with the requirements of Item 402(u) of Regulation S-K. For 2023, our last completed fiscal year:
Based on this information, for 2023 the ratio of CEO annual total compensation to that of the median identified employee is 11:1. This pay ratio is a reasonable estimate calculated in a manner consistent with SEC rules based on our payroll and employment records and the methodology described below. Methodology for Determining Our Median Employee The Company used our employee population data as of December 31, 2021 as the reference date for identifying our median employee. To identify the median employee from our employee population, we calculated each employee’s 2021 base salary, actual amount paid under our short-term incentive compensation program and commission earned in 2021 and grant-date fair value of equity awards granted in 2021. In identifying the median employee, we annualized the compensation of all full-time employees who were new hires in 2021 or were on a leave of absence in a portion of 2021. As there have been no changes that would materially impact our pay ratio analysis, as permitted by SEC rules, we are using the same individual identified in 2021 for our 2023 pay ratio analysis. Annual Total Compensation With respect to the annual total compensation of the employee who represents our median compensated employee, we calculated such employee’s compensation for 2023 in accordance with the requirements of Item 402(c)(2)(x) of Regulation S-K. We used the amount reported in the “Total” column of our 2023 Summary Compensation 35 Pay-Versus-Performance Disclosure As required by Item 402(v) of Regulation S-K, we are providing the following information about the relationship between the compensation actually paid (or “CAP”) to our named executive officers and required financial performance measures. For a more complete explanation of “pay for performance” philosophy and how executive compensation aligns with our performance, please refer to “Executive Compensation – Compensation Discussion and Analysis.” Pay-Versus-Performance Table
Equity Award Valuations – Alan H. Auerbach: Equity award fair values are calculated in accordance with FASB ASC Topic 718. The closing stock price as of December 31, 2022 was $4.23 and at December 31, 2023 was $4.33. The material Black-Scholes assumptions varied as follows: expected term between 3.21 and 5.85, volatility between 75.51% and 86.58%, risk-free rate between 3.81% and 3.95%, and expected dividend yield of 0.00%.
36
Equity Award Valuations – Other named executive officers: Equity award fair values are calculated in accordance with FASB ASC Topic 718. The closing stock price as of December 31, 2022 was $4.23 and at December 31, 2023 was $4.33. The material Black-Scholes assumptions varied as follows: expected term between 3.21 and 5.97, volatility between 75.51% and 86.58%, risk-free rate between 3.81% and 3.95%, and expected dividend yield of 0.00%.
Tabular List of Financial Performance Measures The most important financial and non-financialperformance measures that link CAP to our performance are as follows:
Additional information about each of these performance measures and the role of our performance in each of these measures in determining our executive compensation are discussed in greater detail in “Executive Compensation – Compensation Discussion and Analysis.” Analysis of Information Presented in Pay-Versus-Performance Table As described in more detail in “Executive Compensation – Compensation Discussion and Analysis,” our executive compensation program reflects a variable “pay-for-performance” philosophy. While over the years we have used different performance measures to align executive compensation with our performance, all of these performance measures are not presented in the Pay-Versus-Performance Table. Moreover, while we generally seek to prioritize long-term performance as our primary incentive for Alan H. Auerbach and our other named executive officers, we do not specifically align our performance measures with CAP (as computed in accordance with Item 402(v) of Regulation S-K) for a particular fiscal year. In accordance with Item 402(v) of Regulation S-K, we are providing the following descriptions of the relationships between the information presented in the Pay-Versus-Performance Table. 37 CAP Compared to Company and Peer Group TSR The following graph sets forth the relationship between (i) the CAP for Alan H. Auerbach and the average CAP for our other NEOs as a group (except Alan H. Auerbach), and (ii) our TSR and the TSR of our peer group, the Nasdaq Biotechnology Index, over the periods presented in the Pay-Versus-Performance Table. 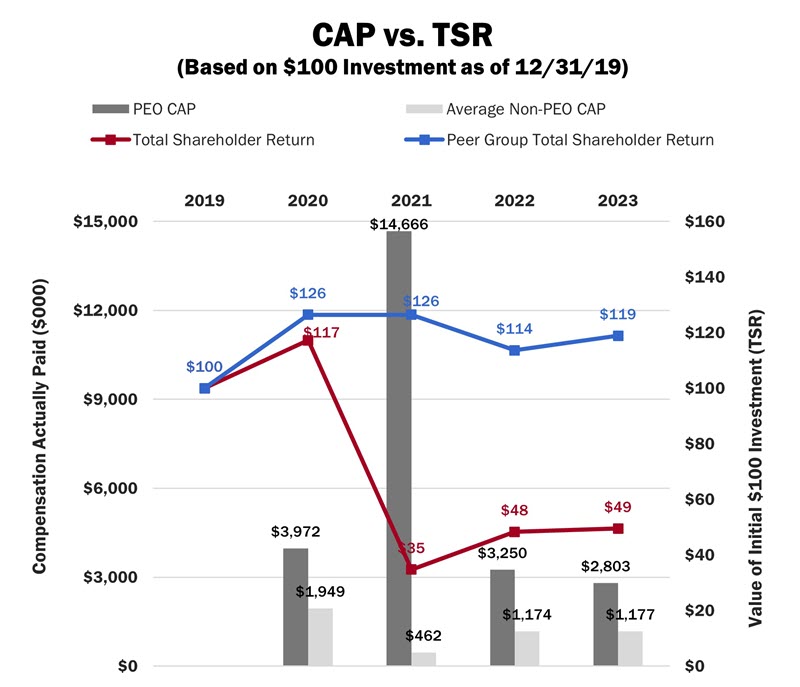 38 CAP Compared to Net Income The following graph sets forth the relationship between (i) the CAP for Alan H. Auerbach and the average CAP for our other NEOs as a group (except Alan H. Auerbach), and (ii) our Net Income over the periods presented in the Pay-Versus-Performance Table. 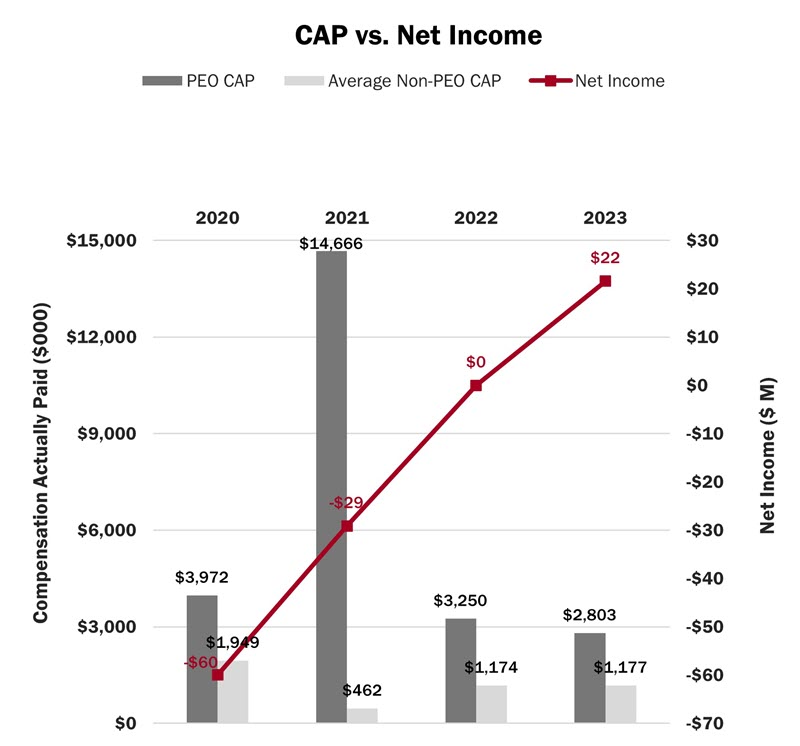 39 CAP Compared to Total Revenue, Less License Revenue The following graph sets forth the relationship between (i) the CAP for Alan H. Auerbach and the average CAP for our other NEOs as a group (except Alan H. Auerbach), and (ii) our Total Revenue, Less License Revenue, over the periods presented in the Pay-Versus-Performance Table. 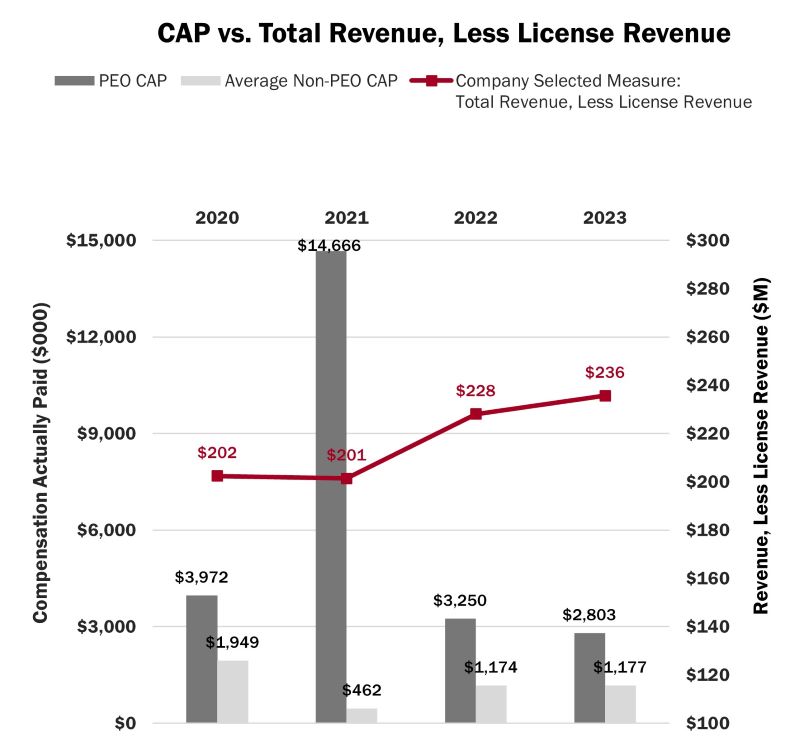 40 Pay-Versus-Performance: Conclusions The Compensation Committee believes in “pay for performance” and has structured our compensation program to reward our executive officers when we are delivering strong results. As part of the 2023 compensation process,our Compensation Committee considers potential risks when reviewing and approving the compensation programs for our Named Executive Officers. In 2023, based on this review, our Compensation Committee concluded that our compensation policies and practices do not incentivize excessive risk-taking that could have a material adverse effect on our Company. The Compensation Committee, comprised of Messrs. Moyes, Miller and Stuglik, has reviewed and discussed the Compensation Discussion and Analysis above with our management. Based on the review and discussions, the Compensation Committee recommended to the Board that the Compensation Discussion and Analysis be included in this proxy statement and incorporated by reference into our Annual Report on Form 10-K for the fiscal year ended December 31, Compensation Committee of the Board of Directors Brian Stuglik (Chair) Michael P. Miller Jay M. Moyes
EQUITY COMPENSATION PLAN INFORMATION The following table sets forth the
In April 2017 our Board of Directors adopted the Inducement Plan. Pursuant to applicable stock exchange rules, stockholder approval of the Inducement Plan is not required as a condition of the effectiveness of the Inducement Plan as the plan will be used to provide equity grants solely to, and in connection with the hiring of, new employees. A description of the principal features of the Inducement Plan is set forth below. In February 2020, our Board of Directors approved an amendment to the Inducement Plan to increase the number of authorized shares by 1,000,000 shares to 2,000,000 shares. In July 2021, our Board of Directors approved an amendment to increase the number of authorized shares by 1,000,000 to 3,000,000 shares. Eligibility and Administration Only certain prospective employees of the Company are eligible to participate in the Inducement Plan. The Inducement Plan is administered by our Compensation Committee. The plan administrator has the authority to make all determinations and interpretations under, prescribe all forms for use with, and adopt rules for the administration of, the Inducement Plan, subject to its express terms and conditions. The plan administrator also sets the terms and conditions of all awards under the Inducement Plan, including any vesting and vesting acceleration conditions. Awards must be approved by the Compensation Committee or a majority of our independent directors and the authority to grant awards under the Inducement Plan may not be delegated. Limitation on Awards and Shares Available The maximum number of shares of our common stock authorized for issuance under the Inducement Plan is 3,000,000 shares. 41 The following types of shares are added back to the available share limit under the Inducement Plan: Shares subject to awards under the Inducement Plan that are forfeited, expire or are settled for cash and shares repurchased by the Company at the same price paid by a participant pursuant to the Company’s repurchase right with respect to
The Inducement Plan
Certain Transactions The plan administrator has broad discretion to equitably adjust the provisions of the Inducement Plan, as well as the terms and conditions of existing and future awards, to prevent the dilution or enlargement of intended benefits and facilitate necessary or desirable changes in the event of certain transactions and events affecting our common stock, such as stock dividends, stock splits, mergers, acquisitions, consolidations and other corporate transactions. In addition, in the event of certain non-reciprocal transactions with our stockholders known as “equity restructurings,” the plan administrator will make equitable adjustments to the Inducement Plan and outstanding awards. In the event of a change in control of our company (as defined in the Inducement Plan), the surviving entity must assume outstanding awards or substitute economically equivalent awards for such outstanding awards; however, if the surviving entity declines to assume or substitute for some or all outstanding awards, then all such awards will vest in full and be deemed exercised (as applicable) upon the transaction. Individual award agreements may provide for additional accelerated vesting and payment provisions. Foreign Participants; Transferability, Repricing and Participant Payments The plan administrator may modify award terms, establish subplans and/or adjust other terms and conditions of awards, subject to the share limits described above, in order to facilitate grants of awards subject to the laws and/or stock exchange rules of countries outside of the United States. With limited exceptions for estate planning, domestic relations orders, certain beneficiary designations and the laws of descent and distribution, awards under the Inducement Plan are generally non-transferable prior to vesting and exercisable only by the participant. The Inducement Plan requires stockholder approval to reprice any award of stock options or SARs (whether through a reduction of the applicable price per share or the cancellation and substitution of such an award with another award when the price per share for such award exceeds the fair market value of the underlying shares). With regard to tax withholding, exercise price and purchase price obligations arising in connection with awards under the Inducement Plan, the plan administrator may, in its discretion, accept cash or check, shares of our common stock that meet specified conditions, a “market sell order” or such other consideration as it deems suitable. 42 Stockholder Approval; Plan Amendment and Termination
Pursuant to applicable stock exchange rules, stockholder approval of the Inducement Plan was not required as a condition of the effectiveness of the Inducement Plan. The Board of Directors may amend Following is the report of the Audit Committee with respect to the Company’s audited Responsibilities. The Audit Committee operates under a written charter adopted by the Board. The role of the Audit Committee is to oversee the Company’s financial reporting process on behalf of the Board. The Company’s management has the primary responsibility for the Company’s financial statements as well as its financial reporting process and principles, internal controls and disclosure controls. Management is responsible for the preparation, presentation and integrity of the Company’s financial statements. Management is also responsible for establishing and maintaining internal controls over financial reporting (as defined in Exchange Act Rule 13a-15(f)) and for evaluating the effectiveness of those internal controls and for evaluating any changes in those controls that will, or Review with Management and Independent Registered Public Accountants. The Audit Committee has reviewed and discussed the audited financial statements for the fiscal year ended December 31, 2023 with management of the Company and with the Company’s independent registered public accounting firm, The Audit Committee has received and reviewed the written disclosures and the letter from Based on the review and discussions referred to above, the Audit Committee recommended to the Board that the audited financial statements be included in the Annual Report on Form 10-K for the year ended December 31, Audit Committee of the Board of Directors Jay M. Moyes Allison Dorval Troy E. Wilson
The following table presents fees for professional services provided
This category includes fees associated with our annual audit and the reviews of our quarterly reports on Form This category includes fees
Pre-Approval Policies and Procedures
Our Audit Committee has pre-approved the
43 The Audit Audit Committee may delegate to one or more designated members of the Audit Committee the authority to grant pre-approvals, provided such approvals are presented to the Audit Committee at a subsequent meeting. If the Audit Committee elects to establish pre-approval policies and procedures regarding non-audit services, the Audit Committee must be informed of each non-audit service provided by the independent auditor. Audit committee pre-approval of non-audit services (other than review and attestation services) also will not be required if such services fall within available exceptions established by the SEC. RATIFICATION OF SELECTION OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM The Audit Committee If the stockholders fail to ratify the selection of OUR BOARD UNANIMOUSLY RECOMMENDS A VOTE
(“SAY-ON-PAY VOTE”) As required by As described in further detail under Proposal 4 of this proxy statement, at this annual meeting, our stockholders are being asked to indicate how frequently they believe we should seek a say-on-pay vote (the “frequency vote”). Our Board has recommended that our stockholders vote for a frequency of one year. We currently expect our next say-on-pay vote (after the vote on this Proposal 3 We encourage our stockholders to review the “Executive Compensation” section of As an advisory approval, this proposal is not binding upon us or
“RESOLVED, that the stockholders
OUR BOARD UNANIMOUSLY RECOMMENDS A VOTE ADVISORY VOTE REGARDING THE FREQUENCY OF FUTURE SAY-ON-PAY VOTES (“FREQUENCY VOTE”) As required by Section 14A(a)(1) of the Exchange Act, our stockholders may indicate how frequently they believe we should seek an advisory vote on the compensation of our Named Executive Officers. We are seeking an advisory, non-binding determination from our stockholders as to the frequency with which our stockholders have an opportunity to provide an advisory approval of the compensation of our Named Executive Officers. Stockholders are not being asked to approve or disapprove our Board’s recommendation, but rather vote for a frequency of one, two or three years, or abstain from voting. 44 We recommend that our stockholders select a frequency of one year, or an annual vote. We believe that this frequency is appropriate because it will continue to enable our stockholders to vote, on an advisory basis, on the most recent executive compensation information that is presented in our proxy statement, leading to a more meaningful and coherent communication between the Company and our stockholders on the compensation of our Named Executive Officers. An annual advisory vote on executive compensation is consistent with our policy of seeking regular input from, and engaging in discussions with, our stockholders on corporate governance matters and our executive compensation philosophy, policies and practices. OUR BOARD UNANIMOUSLY RECOMMENDS THAT STOCKHOLDERS VOTE FOR “1 YEAR” AS THE FREQUENCY OF FUTURE ADVISORY VOTES ON THE COMPENSATION OF THE COMPANY’S NAMED EXECUTIVE OFFICERS. APPROVAL OF
On
The 2011 Plan Amendment is subject to stockholder approval. The summaries of the 2011 Plan and the 2011 Plan Amendment are qualified in their entirety by reference to the 2011 Plan, as amended prior to the Plan Amendment, and the 2011 Plan Amendment, both of which are attached hereto as Appendices A and B, respectively. Proposed Share Reserve Increase and Shares Available for Issuance. We are asking our stockholders to approve the 2011 Plan Amendment because we believe the availability of an adequate reserve of shares under the 2011 Plan is important to our continued growth and success. The purpose of the 2011 Plan is to assist us in attracting, motivating and retaining selected individuals who will serve as our employees, directors and consultants, whose judgment, interest and special effort is critical to the successful conduct of our operation. We believe that the equity-based awards to be issued under the 2011 Plan will motivate recipients to offer their maximum effort to the Company and help focus them on the creation of long-term value consistent with the interests of our stockholders. We believe that grants of equity-based awards are necessary to enable us to continue to attract and retain top talent, as providing equity-based awards is critical to achieving success as we compete for talent in an industry in which equity compensation is market practice and is expected by many existing personnel and prospective candidates. If the 2011 Plan Amendment is not approved, we believe our recruitment and retention goals will be adversely affected. The 2011 Plan Amendment increases the aggregate number of shares available under the 2011 Plan by 3,000,000 shares to a total of 17,529,412 shares of common stock. To the extent we grant any awards under the 2011 Plan between April 1, 2024 and the date of this annual meeting, the available share reserve under the Amended 2011 Plan will be reduced from 3,712,912 shares (i.e., the remaining available reserve as of April 1, 2024 (712,912 shares) plus 3,000,000 shares) by the number of shares that we grant under the 2011 Plan during such period. We expect to register the additional shares authorized under the Amended 2011 Plan on a Registration Statement on Form S-8 within a reasonable period of time following stockholder approval and prior to the issuance of any such shares. The table below presents information about the number of shares that were subject to outstanding equity awards under the 2011 Plan and our Inducement Plan, under which we may grant equity grants solely to, and in connection with the hiring of, new employees, and the shares remaining available for issuance under each plan, each at December 31, 2023. As of April 1, 2024, we had 712,912 shares remaining available for issuance under the 2011 Plan after giving effect to the annual awards granted in 2024 in the aggregate amount of 1,482,375 shares. The 2011 "Plan" and the Inducement Plan are the only equity incentive plans we currently have in place. The table below does not include information about the number of shares exercisable pursuant to the Auerbach Warrant that is reflected in the Equity Compensation Plan Information Table on page 41.
45 Background of Reasons for and the Determination of Shares Under the 2011 Plan Amendment. In its determination to approve the increase in the number of shares authorized under the 2011 Plan, the Board and Compensation Committee were primarily motivated by a desire to ensure the Company has an available pool of shares from which to grant long-term equity-based incentive awards, which the Board and Compensation Committee believe is a primary incentive and retention mechanism. The Board and Compensation Committee considered the following key factors:
In light of the factors described above, the Board believes the proposed increase to the share reserve is reasonable with respect to potential equity dilution and provides a significant incentive for officers, employees, non-employee directors and consultants to increase the value of the Company for all stockholders. Stockholder Approval Stockholder approval of the 2011 Plan Amendment is necessary in order for us to meet the stockholder approval requirements of the principal securities market on which shares of our common stock are traded, and to grant stock options that qualify as ISOs, as defined under Section 422 of the Code. If this Proposal four is adopted, a maximum of 17,529,412 shares of common stock will be reserved for issuance under the Amended 2011 Plan, all of which may be granted as ISOs pursuant to Section 422 of the Code. In addition, we will continue to be able to grant ISOs until March 31, 2034. If stockholders do not approve this Proposal five, the proposed additional shares will not become available for issuance under the 2011 Plan, and we will not be able to continue to grant ISOs after April 1, 2031. Material Terms of the Amended 2011 Plan The material terms of the 2011 Plan, as proposed to be amended by the 2011 Plan Amendment, are summarized below. The following summary is qualified in its entirety by reference to the full text of the 2011 Plan, as amended prior to the 2011 Plan Amendment, and the 2011 Plan Amendment, both of which are attached hereto as Appendices A and B, respectively. Eligibility and Administration. Our employees, consultants and directors are eligible to receive awards under the Amended 2011 Plan. Currently, at April 1, 2024, approximately 183 employees, seven non-employee directors and no consultants are eligible to participate in the Amended 2011 Plan. The Amended 2011 Plan is administered by our Board with respect to awards to non-employee directors and by our Compensation Committee with respect to other participants, each of which may delegate its duties and responsibilities to committees of our directors and/or officers (referred to collectively as the 2011 Plan administrator), subject to certain limitations that may be imposed under Section 16 of the Exchange Act and/or stock exchange rules, as applicable. The 2011 Plan administrator has the authority to make all determinations and interpretations under, prescribe all forms for use with, and adopt Limitation on Awards and Shares Available. If our stockholders approve this proposal, the aggregate number of shares of our common stock that will be available for issuance under awards granted pursuant to the Amended 2011 Plan will equal 17,529,412, all of which may be granted as ISOs pursuant to Section
46 Shares subject to awards under the Amended 2011 Plan that are forfeited, expire or are settled for cash may be used again for new grants under the Amended 2011 Plan. The Awards granted under the Amended 2011 Plan upon the assumption of, or in substitution for, awards authorized or outstanding under a qualifying equity plan maintained by an one participant pursuant to the Amended 2011 Plan during any calendar year is 500,000 shares and the maximum amount that may be paid in cash pursuant to the Amended 2011 Plan to any one participant during any calendar year period will be $2,000,000. The Awards. The Amended 2011 Plan provides for the grant of stock options, including ISOs and non-qualified stock options, or NSOs, restricted stock, dividend equivalents, stock payments, RSUs, deferred stock, performance shares, other incentive awards, SARs, and performance awards (including cash awards). Certain awards under the Amended 2011 Plan may constitute or provide for a deferral of compensation, subject to Section 409A of the Code, which may impose additional requirements on the terms and conditions of such awards. All awards under the Amended 2011 Plan are or will be set forth in award agreements, which detail the terms and conditions of the awards, including any applicable vesting and payment terms and post-termination exercise limitations. Awards other than cash awards are generally settled in shares of our common stock, but the 2011 Plan administrator may provide for cash settlement of any award. A brief description of each award type follows.
47 Certain Transactions. The 2011 Plan administrator has broad discretion to equitably adjust the provisions of the Amended 2011 Plan, as well as the terms and conditions of existing and future awards, to prevent the dilution or enlargement of intended benefits and facilitate necessary or desirable changes in the event of certain transactions and events affecting our common stock, such as stock dividends, stock splits, mergers, acquisitions, consolidations and other corporate transactions. In addition, in the event of certain non-reciprocal transactions with our stockholders known as “equity restructurings,” the 2011 Plan administrator will make equitable adjustments to the Amended 2011 Plan and outstanding awards. In the event of a change in control of our company (as defined in the Amended 2011 Plan), the surviving entity must assume outstanding awards or substitute economically equivalent awards for such outstanding awards; however, if the surviving entity declines to assume or substitute for some or all outstanding awards, then all such awards will vest in full and be deemed exercised (as applicable) upon the transaction. Individual award agreements may provide for additional accelerated vesting and payment provisions. Foreign Participants, Transferability, Repricing and Participant Payments. The 2011 Plan administrator may modify award terms, establish subplans and/or adjust other terms and conditions of awards, subject to the share limits described above, in order to facilitate grants of awards subject to the laws and/or stock exchange rules of countries outside of the United States. With limited exceptions for estate planning, domestic relations orders, certainbeneficiary designations and the laws of descent and distribution, awards under the Amended 2011 Plan are generally non-transferable prior to vesting and exercisable only by the participant. The Amended 2011 Plan requires stockholder approval to reprice any award of stock options or stock appreciation rights (whether through a reduction of the applicable price per share or the cancellation and substitution of such an award with another award when the price per share for such award exceeds the fair market value of the underlying shares). With regard to tax withholding, exercise price and purchase price obligations arising in connection with awards under the Amended 2011 Plan, the 2011 Plan administrator may, in its discretion, accept cash or check, shares of our common stock that meet specified conditions, a “market sell order” or such other consideration as it deems suitable. Plan Amendment and Termination. Our Board may amend or terminate the Amended 2011 Plan at any time; however, except in connection with certain changes in our capital structure, stockholder approval will be required for (i) any amendment that increases the number of shares available under the Amended 2011 Plan and (ii) any amendment that reduces the price per share of any outstanding option or stock appreciation right granted under the Amended 2011 Plan or that cancels any stock option or stock appreciation right in exchange for cash or another award when the option or stock appreciation right price per share exceeds the fair market value of the underlying shares. If our stockholders approve this proposal, then after March 21, 2034, no ISOs may be granted; if the proposal is not approved then we will not be able to New Plan Benefits Except with respect to grants of restricted stock unit awards that will be granted to each non-employee director serving on our board at this annual meeting, which is shown in the
Equity Awards Made as of April 1, 2024 The following table sets forth summary information concerning the number of shares of our common stock subject to option grants, restricted stock unit grants and performance share grants that have been made under the 2011 Plan to our named executive officers, directors and employees as of April 1, 2024 since its inception. These numbers do not reflect currently outstanding awards. 48 Certain awards set forth in this table for the named executive officers were granted in 2023 and therefore also are included in the Summary Compensation Table and in the Grants of Plan-Based Awards Table set forth in this proxy statement and are not additional awards. Certain awards set forth in this table for the non-employee directors were granted in 2023 and therefore also are included in the Director Compensation Table set forth in this proxy statement and are not additional awards.
Material U.S. Federal Income Tax Consequences The following is a general summary under current law of the principal United States federal income tax consequences related to awards under the Amended 2011 Plan. This summary deals with the general federal income tax principles that apply and is provided only for general information. Some kinds of taxes, such as state, local and foreign income taxes and federal employment taxes, are not discussed. This summary is not intended as tax advice to participants, who should consult their own tax advisors. Non-QualifiedStock Options. If an optionee is granted an NSO under the Amended 2011 Plan, the optionee should not have taxable income on the grant of the option. Generally, the optionee should recognize ordinary income at the time of exercise in an amount equal to the fair market value of the shares acquired on the date of exercise, less the exercise price paid for the shares. The optionee’s basis in the common stock for purposes of determining gain or loss on a Incentive Stock Options. A participant receiving ISOs should not recognize taxable income upon grant. Additionally, if applicable holding period requirements are met, the participant should not recognize taxable income at the time of
Other Awards. The current federal income tax consequences of other awards authorized under the Amended 2011 Plan generally follow certain basic patterns: SARs are taxed and deductible in substantially the same manner as NSOs; nontransferable restricted stock subject to a substantial risk of forfeiture results in income recognition equal to the excess of the fair market value over the price paid, if any, only at the time the restrictions lapse (unless the recipient elects to accelerate recognition as of the Excess Parachute Payments. Section 280G of the 49 Application of Section 409A of the Code. Section 409A of the Code imposes an additional 20% tax and Securities Law Matters We expect to register the additional shares authorized under the 2011 Plan on a Registration Statement on Form S-8 within a reasonable period of OUR BOARD UNANIMOUSLY RECOMMENDS A VOTE Policies and Procedures for Review, Approval or Ratification of Transactions with Related Persons Under our written Related Party Transactions Policy and Procedures, a related party transaction (as defined below) may be consummated or may continue only if the For the purposes of our policy, a “related party transaction” is a transaction, arrangement or relationship (or any series of similar transactions, arrangements or relationships) in which the Company (including any of its subsidiaries) was, is or will be a participant and the amount involved exceeds $120,000, and in which any related party (as defined below) had, has or will have a direct or indirect interest. A “related party” includes (i) any person who is, or at any time since the beginning of the Company’s last fiscal year was, a director or executive officer of the Company or a nominee to become a director of the Company; (ii) any person who is known to be the beneficial owner of more than 5% of any class of the Company’s voting securities; (iii) any immediate family member of any of the foregoing persons, which means any child, stepchild, parent, stepparent, spouse, sibling, mother-in-law, father-in-law, son-in-law, daughter-in-law, brother-in-law, or sister-in-law of the director, executive officer, nominee or more than 5% beneficial owner, and any person (other than a tenant or employee) sharing the household of such director, executive officer, nominee or more than 5% beneficial owner; and (iv) any firm, corporation or other entity in which any of the foregoing persons is employed or is a general partner or principal or in a similar position or in which such person has a 5% or greater beneficial ownership interest. Delinquent Section 16(a) Section 16(a) of the Exchange Act requires our directors and officers, and persons who beneficially own more than ten percent (10%) of our Stockholder Proposals and Nominations Proposals Pursuant to Rule 14a-8. Pursuant to Rule 14a-8 under the Exchange Act, stockholders may present proper proposals for inclusion in the proxy statement and for consideration at our next annual meeting of stockholders. To be eligible for inclusion in the Proposals and Nominations Pursuant to Our Fourth Amended and Restated Bylaws. Proposals Pursuant to Rule 14a-19. In addition to satisfying the foregoing requirements under our Bylaws, to comply with the universal proxy rules, stockholders who intend to solicit proxies in support of director nominees other than our nominees must provide notice that sets forth the information required by Rule 14a-19 under the Exchange Act no later than April 19, 2025. We intend to file a Proxy Statement and WHITE proxy card with the SEC in connection with our solicitation of proxies for our 2025 annual meeting of stockholders. Stockholders may obtain our Proxy Statement (and any amendments and supplements thereto) and other documents as and when filed by us with the SEC without charge from the SEC’s website at: www.sec.gov. 50 Householding of Proxy Materials The SEC has adopted rules that permit companies and intermediaries (such as banks and brokers) to satisfy the delivery requirements for notices of annual meetings, proxy statements and annual reports with respect to two or more stockholders sharing the same address by delivering a single proxy statement addressed to those stockholders. This process, which is commonly referred to as “householding,” potentially means extra convenience for stockholders and cost savings for companies. This year, a single notice of the annual meeting of stockholders, or copy of the proxy statement and annual report, will be delivered to multiple stockholders sharing an address unless contrary instructions have been received from the affected stockholders. Once you have received notice from your bank or broker that it will be householding communications to your address, householding will continue until you are notified otherwise or until you revoke your consent. If, at any time, you no longer wish to participate in householding and would prefer to receive a separate proxy statement and annual report, please notify your bank or broker, direct your written request to Puma Biotechnology, Inc., 10880 Wilshire Boulevard, Suite 2150, Los Angeles, CA 90024, Attention: Investor Relations, or contact Investor Relations by telephone at (424) 248-6500. Stockholders who currently receive multiple copies of the proxy statement at their address and would like to request householding of their communications should contact their bank or broker. Notwithstanding anything to the contrary set forth in any of our previous filings under the Securities Act or the Exchange Act, which might incorporate future filings made by us under those statutes, the Compensation Committee Report, This Proxy Statement contains forward-looking statements within the meaning of Section 21E of the Securities Exchange Act of 1934, as amended, including, but not limited to, statements related to risks associated with our compensation programs. All forward-looking statements included in this Proxy Statement involve risks and uncertainties that could cause the Company’s actual results to differ materially from the anticipated results and expectations expressed in these forward-looking statements. These statements are based on current expectations, forecasts and assumptions, and actual outcomes and results could differ materially from these statements due to a number of factors, which include, but are not limited to, the Company’s dependence on the commercial success of its product candidates, the Company’s history of operating losses and its expectation that it will continue to incur losses for the foreseeable future; risks and uncertainties related to the Company’s ability to achieve or sustain profitability; the Company’s ability to predict its future prospects and forecast its financial performance and growth; failure to obtain sufficient capital to fund the Company’s operations; the effectiveness of sales and marketing efforts; the Company’s ability to obtain regulatory approval of NERLYNX outside the United States; the Company’s licensees’ ability to obtain FDA approval or other regulatory approvals in the United States or elsewhere for other indications for neratinib or other product candidates; the challenges associated with conducting and enrolling clinical trials; the risk that the results of clinical trials may not support the Company’s drug candidate claims; the Company’s ability to compete against other companies and research institutions; the risk that physicians and patients may not accept or use the Company’s products; the Company’s reliance on third parties to conduct its clinical trials and to formulate and manufacture its drug candidates; risks pertaining to litigation, including the Company’s estimates for damages that it may be required to pay; the protection for its intellectual property; the Company’s ability to attract and retain key personnel; the Company’s dependence on licensed intellectual property; any adverse impact on the Company’s business or the global economy and financial markets, generally; and the other risk factors disclosed in the periodic and current reports filed by the Company with the Securities and Exchange Commission from time to time, including the Company’s Annual Report on Form 10-K for the fiscal year ended December 31, 2023. Readers are cautioned not to place undue reliance on these forward-looking statements, which speak only as of the date hereof. The Company assumes no obligation to update these forward-looking statements, except as required by law. As of the date of this proxy statement, the Board knows of no other business that will be presented for consideration at the
51 PUMA BIOTECHNOLOGY, INC.
ARTICLE 1. The ARTICLE 2. Wherever the following terms are used in the Plan they shall have the meanings specified below, unless the context clearly indicates otherwise. The singular pronoun shall include the plural where the context so indicates.
52
Notwithstanding the foregoing, if a Change in Control constitutes a payment event with respect to any Award which provides for the deferral of compensation that is subject to Section 409A of the Code, to the extent required to avoid the imposition of additional taxes under Section 409A of the Code, the transaction or event described in subsection (a), (b), (c) or (d) with respect to such Award shall only constitute a Change in Control for purposes of the payment timing of such Award if such transaction also constitutes a “change in control event,” as defined in Treasury Regulation Section 1.409A-3(i)(5). Consistent with the terms of this Section 2.8, the Administrator shall have full and final authority to determine conclusively whether a Change in Control of the Company has occurred pursuant to the above definition, the date of the occurrence of such Change in Control and any incidental matters relating thereto.
53
54
55
The Administrator, in its sole discretion, shall determine the effect of all matters and questions relating to Terminations of Service, including, without limitation, the question of whether a Termination of Service has occurred, whether any Termination of Service resulted from a discharge for Cause and all questions of whether any particular leave of absence constitutes a Termination of Service; provided, however, that, with respect to Incentive Stock Options, unless the Administrator otherwise provides in the terms of any Program, Award Agreement or otherwise, a leave of absence, change in status from an employee to an independent contractor or other change in the employee-employer relationship shall constitute a Termination of Service only if, and to the extent that, such leave of absence, change in status or other change interrupts employment for the purposes of Section 422(a)(2) of the Code. For purposes of the Plan, a Participant’s employee-employer relationship or consultancy relationship shall be deemed to be terminated in the event that the Affiliate employing or contracting with such Participant ceases to remain an Affiliate following any merger, sale of stock or other corporate transaction or event (including, without limitation, a spin-off). 56 ARTICLE 3.
57 ARTICLE 4.
58 ARTICLE 5.
59 ARTICLE 6.
60 ARTICLE 7.
61 ARTICLE 8.
62 ARTICLE 9.
63 ARTICLE 10. STOCK APPRECIATION RIGHTS
64 ARTICLE 11. ADDITIONAL TERMS OF AWARDS
65
66 ARTICLE 12.
67
68 ARTICLE 13.
69
70
71 SIXTH AMENDMENT TO PUMA BIOTECHNOLOGY, INC. 2011 INCENTIVE AWARD PLAN This Sixth Amendment (“Sixth Amendment”) to the Puma Biotechnology, Inc. 2011 Incentive Award Plan, as amended (the “Plan”),
AMENDMENT The Plan is hereby amended
“2.19 “Effective Date” shall mean June 18, 2024, provided, however, that solely for purposes of the last sentence of Section 13.1, the Effective Date shall be the date on which the Plan (as amended by the
72
  | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||